Introduction
Prefilled glass syringes (PFS) are increasingly becoming a container of choice for storing and administering therapeutic protein products to patients [1]. The PFS is a convenient and reliable system for injection compared with the more traditional method of transferring, measuring, and delivering a dose from a vial containing liquid or reconstituted lyophilized powder. In addition, the PFS can deliver a controlled volume minimizing drug waste associated with vial overfills.
PFS is a primary container and its compatibility with the drug needs to be addressed for ensuring patient safety and drug quality [2, 3]. Safety of PFS is the responsibility of drug manufacturers and understanding PFS extractables/leachables is important for assessing syringe compatibility. Identification and quantitation of extracted/leached chemicals is critical for assessing toxicological and drug quality risks. A high extractable/leachable risk assessment may simply disqualify a syringe and eliminate the need to conduct further resource intensive qualifications assessments such as stability, particle formation, container closure integrity, break loose extrusion, and others. Understanding extractable/leachable contributes to the PFS knowledge space and may also serve to create collaboration opportunities with suppliers for reducing/eliminating leachables and improving PFS systems [4]. This paper describes extractables/leachables from syringes and its implication on biological products.
Extractables and Leachables
Extractables are chemicals that migrate from the product-contact component into a solvent at accelerated conditions (such as heat, time, pH, ionic strength, organic solvent content). Leachable are chemicals that migrate from the product-contact component into a formulated drug during normal storage/usage conditions.
Information regarding leached chemicals from components is not known or readily available from the supplier. Goals of extractable studies are to generate, identify, and predict leachables. Extraction under appropriate solvent, temperature, and exposure conditions can generate representative leachables and in large enough quantities to facilitate structure elucidation analysis [5, 6]. Assembled syringes and individual components can be extracted with various solvents (water and water/organic mixtures, pH, ionic strength) at elevated temperatures for a specified duration [5-8]. The identified extractables provide insight on predicting leachables. Leachables can be identical or a subset (oxidation, derivative, etc.) of extractables, or can form adduct products with other leachables, excipients, and proteins.
Extractables information is beneficial since analyzing leachables directly in the formulated drug is challenging due to low leachable concentrations (ppm/ppb range) and matrix inference from protein and excipients. Also categorizing foreign chemicals in drug products as syringe component or drug process impurity related is inconclusive when analyzing leachables directly. Extractables data can determine the source of the leachables.
Analytical Techniques
Analytical characterization and quantitation techniques associated with organic and inorganic chemicals are commonly used to analyze extractable/leachables [9]. Gas chromatography mass spectrometry (GCMS), solid phase micro extraction (SPME)-GCMS, liquid chromatography mass spectrometry (LCMS), high performance liquid chromatography (HPLC), and nuclear magnetic resonance (NMR) spectroscopy techniques are used to analyze unknown organic extractable compounds. Inductively coupled plasma mass spectrometry (ICPMS) can be used to identify and quantitate inorganic metallic elements. Evaporative light scattering detection (ELSD) can be supplemental to HPLC and suitable for oligomers/polymers and non-chromophores molecule analysis.
PFS Components, Leachables, and Implications
Sources of leachables can be from the PFS components (glass barrel, stainless steel hypodermic needle, rubber needle shield, silicone oil lubricants, and fluoropolymer coated rubber plunger) and less obvious sources are from [8, 10] residues from processing tools [11-13] and additives for attaching the needle to the barrel [4]. The impact of leachables on therapeutic proteins can be related to aggregation, particle formation, and/or product quality issues such as reaction with the formulation or protein. Many of these negative attributes may not be observed in non PFS containers possibly due to short contact times during the uptake and delivery from vials using non PFS. However, the shelf-life for biological therapeutics in PFS can be greater than one year allowing leachables to accumulate and potentially interact with drug formulations [14].
The impact from leachables may not be universally observed across all formulated products in a pipeline since formulation factors (pH, ionic strength, surfactants, excipients, etc.) and/or protein attributes may vary.
Glass
Type I borosilicate glass is commonly used to make prefilled syringes and contains various inorganic oxides such as boron, silicon, calcium, sodium, potassium, iron, and aluminum [15]. These inorganic oxides may not pose a direct toxicological risk but migration of glass components from ion-exchange, glass dissolution, pitting, stress, surface layer exfoliation, weathering, and/or erosion corrosion effects may lead to particle formation. [7,15].
Rubber Materials (Plunger and Needle Shield)
The plunger and needle shield are composed of rubber related materials. The product contact surface of the plunger is laminated with a fluoropolymer type film and the needle shield is unlaminated. Contact between the formulated drug with the plunger and shield may result in leached rubber additives or bromine related compounds. Leached rubber agents have been proposed to cause adverse patient effects [10].
Hypodermic Needle
The attached hypodermic needle is made of stainless steel. Inorganic metals used in the stainless steel formulation such as Fe, Cr, Mn, Ni, and Mo may leach from the needle. Based on our experience, metals leach at low levels and are not a direct toxicological concern. However under specific conditions and concentrations, metals such as Mn may catalyze oxidization reactions on proteins [16].
Adhesive
UV activated adhesives composed of organic and oligmeric/polymeric materials are commonly used to bind the stainless steel hypodermic needle to the glass barrel [4]. Controlling the adhesive formulation, application, activation/curing, residues, and clean up processes are important steps for preventing/reducing these materials from coming in contact and leaching into the drug formulation.
While evaluating PFS models from various vendors, several organic, oligomeric, and acrylate related materials consistent to those used in the adhesive industry were extracted and characterized [17]. These adhesive residues did leach into various formulated protein products and did not appear to induce precipitation, aggregation, or particles. The impact on drug quality was a concern since acrylates are reactive toward protein under certain conditions [18]. Evaluation of a formulated protein stored in syringes containing endogenous levels of adhesives at 37°C for 45 days led to adduct formation. Irreversible addition of acrylates with the formulated protein was observed at multiple lysine, histidine, and N-terminus sites with 0.02% of amino acid sites being modified [19].
Silicone Oil
Silicone oil is applied to coat the barrel, plunger, and needle exterior. The most common form of silicone oil used in medical applications is polydimethylsiloxane and functions as a lubricant allowing the plunger to glide smoothly within the barrel to expel the drug. As the demand for prefilled syringe and automated injection devices increases, so does the importance of understanding silicone oil. Functionally, silicone oil is not a major concern for manual injections since a nurse or doctor is capable of applying the necessary force to push the plunger to the end point. However, a spring can only provide a fixed amount of force and any unanticipated friction may cause the plunger to stall before complete drug delivery.
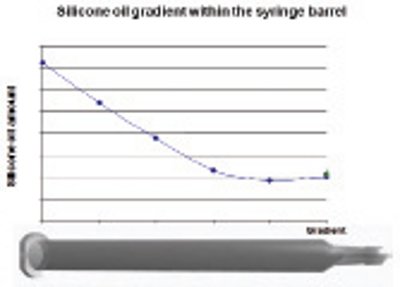
Figure 1-Silicone oil gradient in an empty PFS [23].
Although silicone oil is considered inert and insoluble in water, it may interact with the formulation causing protein aggregation [20,21], droplets, and particles [22]. Ideally the silicone oil application process should balance the need to minimize the undesired drug quality attributes and to provide sufficient lubrication. The silicone oil distribution is non-uniformly distributed with the least amount located near the needle end of the syringe (Figure 1) [23].

Figure 2- Tungsten oxides inside the syringe near the hypodermic needle-barrel zone [24].
Syringe Tool – Tungsten Pin
Less obvious sources of PFS extractables are contamination from tools that were used to manufacture and process syringes. Glass syringes are made from cutting and molding glass tubing at high temperatures. Heated glass is in contact with various processing tools during the process. The barrel’s inner channel cavity for holding a stainless steel needle is formed at approximately 1,200 °C using a tungsten pin. Tungsten is commonly used due its heat resistance and relative high melting temperature. However at temperatures greater than 150 °C, tungsten oxidizes in the presence of air and leaves a white tungsten containing residue within the syringe (Figure 2). These residues may survive the syringe washing step and may contact the drug upon syringe filling and storage. The tungsten oxide residues did not cause protein precipitation above pH 5, but caused protein aggregation below pH 5 [12, 24]. Below pH 5, ppm low amounts (parts per million, ppm) of tungsten oxide formed large tungstate polyanions which did aggregate protein at low ppm levels.

Figure 3- Polymeric pin and a polymeric pin inserted into a syringe.
Syringe Tool – Polymeric Pin
Polymeric nylon pins are used to transport glass syringe barrels on an assembly line. These reusable pins (approximately 0.5 x 6 cm) fit within the hot syringe (Figure 3). Abnormal heat exposure or extensive pin usage may lead to pin wear and tear. Pin residues exposed to heated glass syringes may adhere to the inner syringe wall and survive wash and rinse procedures. During a visual inspection of a filled syringe product a black residue was observed (Figure 4) and later identified by LCMS and FTIR analysis as containing nylon related species [11]. Substances from the black residue did leach into the drug formulation and analysis confirmed the leachables and solid black residue matched those of the nylon pin used by the syringe manufacture. The syringe manufacturer was notified and has implemented measures to prevent future occurrences.

Figure 4-Two black particles observed inside a pre-filled syringe. Larger particle is approximately 300 microns [11].
Conclusion
PFS components and residues from processing tools may leach organic and inorganic chemicals into formulated drugs. Leachable information is often not readily available from the syringe manufacturer prompting drug manufacturers to initiate extractable/leachable investigations. Leachable from PFS may or have contributed to safety concerns related to protein aggregation, particle formation, and toxicological risk factors. Identifying extractables and leachables provide key information enabling safety assessments that address toxicology and drug quality impact for evaluating PFS.
Acknowledgements
Authors would like to thank Joseph Phillips and David Brems for their efforts and useful discussions.
References
- Thompson, I. “New-Generation Auto-Injectors: Completing the Scale of Convenience for Self-injection. Drug Delivery Report. 2005, Autumn/Winter, 47-49.
- Markovic I. Risk Management Strategies for Safety Qualification of Extractable and Leachable Substances in Therapeutic Biologic Protein Products. Am Pharm. Rev. 2009, 12(4) 96-101.
- Guidance for Industry. 1999. Container closure systems for packaging human drugs and biologics. Rockville, MD: US Department of Health and Human Services, Food and Drug Administration.
- Sardella, A. Fine tuning of process parameters for improving biocompatibility of prefillable syringes. Ondrugdelivery. 2010, (January) 18-22
- Jenke, D. R. Evaluation of model solvent systems for assessing the accumulation of container extractables in drug formulations. J. Pharm. Sci. 2001, 224 (1-2), 51–60.
- Jenke, D. R. Linking extractables and leachables in container/closure applications. PDA J. Pharm. Sci. Technol. 2005, 59 (4), 265–281.
- Borchert, S.J.; Ryan, M.M.; Davidson, R.L.; Speed, W. Accelerated extractable studies of borosilicate glass containers. J. Parenter. Sci. and Technol. 1989, 43(2) 67-79.
- Wakankar, A.A.; Wang, J. Y.; Canova-Davis, E.; Ma S.; Schmalzing, D.; Grieco, J.; Milby, T.; Reynolds, T.; Mazzarella, K.; Hoff, E.; Gomez, S.; Martin-Moe, S. On Developing a Process for Conducting Extractable-Leachable Assessment of Components Used for Storage of Biopharmaceuticals. J. Pharm. Sci. 2010, 99(5), 2209–2218.
- Wang, Q. Selection of Analytical techniques for pharmaceutical leachables studies. Am Pharm. Rev. 2005, 8(6) 38-44.
- Jenke, D. Suitability-for-Use Consideration for Prefilled Syringes. Pharm. Technol. 2008, April 1 issue.
- Nashed, Y.; Torraca, G.; Liu, D.; Fujimori, K.; , Zhang, Z.; Wen, Z.; Lee, H. “Identification of an Extraneous Black Particle in a Glass Syringe: Extractables/Leachables Case Study.” PDA J. Pharm. Sci. Tech. 2010, 64, 242-248.
- Jiang, Y. ; Nashed-Samuel, Y. ; Li, C. ; Liu, W. ; Pollastrini, J. ; Mallard, D. ; Wen, ZQ. ; Fujimori. K. ; Pallitto, M. ; Donahue, L . ; Chu, G. ; Torraca, G. ; Vance, A. ; Mire-Sluis, T. ; Freund, E. ; Davis, J. ; Narhi, L. J. Pharm Sci. 2009, 98(12), 4695-710.
- Liu, W.; Swift, R.; Torraca, G. ; Nashed-Samuel, Y. ; Wen, ZQ. ; Jiang, Y. ; Vance, A. ; Mire-Sluis, A. ; Freund, E. ; Davis, J. ; Narhi, L. PDA J. Pharm. Sci. Tech. 2010, 64(1), 11-19.
- Hung G.W.; Nunez L.J.; Autian J. Correlation of kinetic parameters and thermal behavior of segmented polyurethane elastomers with biological responses. J. Pharm. Sci. 1975, 64, 1492–1497.
- Walther, M.; Rupertus, V.; Seemann, C.; Brecht, J.; Hormes, R.; Swift, R.W. Pharmaceutical Vials with Extremely High Chemical Inertness. PDA J. Pharm. Sci. Technol. 2002, 56(3), 124-129.
- Deman, J. M. Principles of food chemistry 3rd edition. (1999) Aspen Publishers. ISBN: 0-8342-1234-X, 131-132.
- Nashed-Samuel, Y. Extractable and Leachable Implications on Biological Products in Prefilled Syringes. PDA/FDA Joint Regulatory Conference, September 13-16, 2010.
- Potter, D. W.; Tran, T. Rates of Ethyl Acrylate Binding to Glutathione and Protein. Toxicology Letters. 1992, 62, 275-285.
- . Liu, D.; Nashed-Samuel, Y.; Bondarenko, P. V.; Brems, D. N.; Ren, D. Interactions Between Therapeutic Proteins and Acrylic Acid Leachate. In preparation.
- Jones, L. S., Kaufmann, A., and Middaugh, C. R. Silicone Oil Induced Aggregation of Proteins. J. Pharm. Sci. 2005, 94(4), 918-927.
- Thirumangalathu, R., Krishnan, S., Ricci, M.S., Brems, D.N., Randolph, T.W., and Carpenter, J. F. Silicone Oil- and Agitation-Induced Aggregation of a Monoclonal Antibody in Aqueous Solution. J. Pharm. Sci. 2009, 98(9), 3167-3181.
- Markovic, I. Challenges Associated with Extractable and/or Leachable Substances in Therapeutic Biologic Protein Products. Am. Pharm. Rev. 2006, 9(6), 20-27.
- Lee, H.; Liu, D.; Fujimori, K.; Perez, L.; Nashed-Samuel, Y. Unpublished results. 2006.
- Lee, H.; Nashed-Samuel, Y.; Fujimori, K.; Liu, D.; Perez, L. Tungsten Leaching from Prefilled Syringes and Impact on Protein Aggregation. Poster presented at the PDA Extractables/Leachables Forum: Confronting Extractables and Leachables Issues in an Evolving Industry; Bethesda, Maryland, November 6–8, 2007.
This article was printed in the January/February 2011 issue of American Pharmaceutical Review - Volume 14, Issue 1. Copyright rests with the publisher. For more information about American Pharmaceutical Review and to read similar articles, visit www.americanpharmaceuticalreview.com and subscribe for free.