Center for Drug Evaluation and Research
Center for Drug Evaluation and Research
Center for Drug Evaluation and Research
This article reflects the views of the authors and should not be construed to represent FDA’s views or policies.
Introduction
Polylactide (PLA)/Poly(lactide-co-glycolide) (PLGA) is widely used in many FDA-approved drug products. There are currently 15 FDA-approved PLA/PLGA-based drug products available on the US market (Table 1). PLA/PLGA-based drug products are designed to reduce dosing frequency and potential drug toxicity. They are also useful for improving patient compliance with a better therapeutic option to treat patients who adhere poorly to frequent oral or injectable medication. However, as these products are generally expensive, the availability of generic PLA/PLGA-based counterparts would make them more affordable, more widely available, and thus benefitting more patients.
Table 1. PLA/PLGA-based drug products that are available in the U.S.
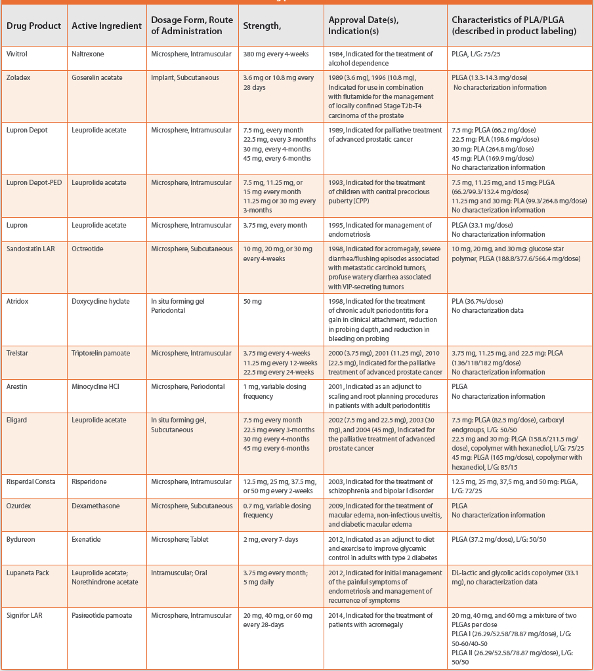
To gain FDA approval, generic drug products must demonstrate pharmaceutical equivalence and bioequivalence to the reference listed drug (RLD). Pharmaceutical equivalence requires that the drug product contains the same active ingredient(s) as the RLD, be identical in strength, dosage form, and route of administration, and that it meets compendial or other applicable standards of strength, quality, purity, and identity. Bioequivalence requires an absence of a significant difference in the rate and extent to which the active ingredient in pharmaceutically equivalent products becomes available at the site of action. For injectable PLA/PLGA-based drug products, the test product should be qualitatively (Q1) and quantitatively (Q2) the same as the RLD to be considered for approval in an Abbreviated New Drug Application (ANDA).1
Differences in PLA/PLGA characteristics can vastly alter the drug release mechanism and release rate. As a random copolymer, the inherent heterogeneity associated with PLA/PLGA makes determination of Q1 sameness of the PLA/PLGA very challenging. Identifying the key characteristics of PLA/PLGA is critical for developing appropriate regulatory standards to determine Q1 sameness of PLA/PLGA and pharmaceutical equivalence between the test and reference products.
Moreover, even with Q1/Q2 sameness, bioequivalence between the test and reference products cannot be ensured, as differences in manufacturing processes may cause differences in physicochemical characteristics of the test product and subsequently affect drug release behavior and bioavailability. In vitro drug release testing in combination with other characterization studies can be used to evaluate differences of the drug product caused by differences in PLA/PLGA properties and/or manufacturing processes. However, to date, compendial or biorelevant in vitro drug release assays and relevant characterization studies for PLA/PLGA-based drug products are still lacking.
To date, the Office of Generic Drugs (OGD) has posted seven product specific recommendations for microsphere products to provide guidance on bioequivalence study design. OGD is working to develop recommendations for other PLA/PLGA-based drug products. Compared to the bioequivalence studies of conventional dosage forms, bioequivalence studies on microspheres are more complicated. Microspheres may have multi-phasic release profiles both in vitro and in vivo, which warrant a carefully designed bioequivalence study to capture significant differences in product performance during each phase.
Currently, no PLA/PLGA-based generic drug products have been approved. This can be attributed to several factors. Adequate characterization of PLA/PLGA needs to be submitted in an ANDA, which may sometimes be challenging. Compendial methods of in vitro release testing of PLA/PLGA-based products are lacking. In addition, the clinical study (i.e., clinical endpoint or pharmacokinetic bioequivalence study) may be a challenge to perform. Recognizing these challenges associated with development of generic PLA/PLGAbased drug products, a regulatory science research program was established under the Generic Drug User Fee Amendments (GDUFA) and is being implemented by OGD to provide new tools for ANDA review and support generic product development.1
GDUFA Regulatory Science Program
Since the enactment of GDUFA in July 2012, OGD has awarded grants and contracts for multiple research projects involving PLA/PLGAbased drug products in various dosage forms, such as microspheres, implants, and in situ gelling systems. Broadly, these projects can be categorized into four areas: (1) development of in vitro-in vivo correlations (IVIVC), (2) development of in vitro release testing (IVRT) methods, (3) characterization of PLA/PLGA, and (4) modeling and simulation of PLA/PLGA-based drug products (Table 2).
Table 2. Research projects involving PLA/PLGA-based drug products

IVIVC of Risperidone Microspheres
It is challenging to develop a reproducible and discriminatory in vitro release method for microspheres, and it is even more challenging to develop an IVIVC for these products. A Level A IVIVC may allow an in vitro release method to be used as a surrogate for bioequivalence studies if any changes occur in formulation and/or manufacturing process post approval. Microspheres often have multiphasic in vitro release and in vivo PK profiles, which makes the deconvolution of in vivo data and correlation with in vitro data very challenging. In addition, the lack of compendial in vitro release testing methods also hinders development of an IVIVC for microspheres.
A study was conducted to explore the possibility of developing an IVIVC for risperidone microspheres that are equivalent in formulation composition but prepared with different manufacturing processes.14 The physicochemical properties of the microspheres were sensitive to minor manufacturing changes, such as solvent systems. Two in vitro release methods (sample-and-separation and USP apparatus 4) were investigated. Compared with the sample-and-separate method, the USP apparatus 4 method showed better discriminatory power for differentiating release profiles of compositionally equivalent microspheres with manufacturing differences. Level A IVIVCs were established and validated for the prepared microspheres based on the in vitro release data obtained using USP apparatus 4. These results indicated that the developed USP apparatus 4 method was capable of discriminating risperidone microspheres that are Q1/Q2 equivalent but with manufacturing differences and predicting their in vivo performance in the investigated rabbit model. It is noted that the developed IVIVCs for risperidone microspheres are based on animal data and may not be fully extrapolated to humans.
A Reproducible Accelerated In Vitro Release Testing Method For Microspheres
A study was also conducted to develop a discriminatory and reproducible accelerated in vitro release method for risperidone microspheres that are Q1/Q2 equivalent, but with different inner structures resulting from differences in manufacturing16. Since real time release testing of microspheres requires extended periods of time, it is beneficial to develop a reproducible accelerated release method, which can correlate with real time release, to provide fast quality assessment and facilitate product development. Two in vitro release methods (sample-and-separation and flow through) were investigated. The results indicated that both methods were able to differentiate risperidone microspheres with different porosities under real time (37°C) and accelerated (45°C) conditions. However, it is worth noting that only the USP apparatus 4 method under the accelerated condition showed good reproducibility for formulations that are highly porous. Therefore, the accelerated release method using USP apparatus 4 may be suitable as a fast quality control for PLGA microspheres (even with porous structures).
A Protocol For Assay Of PLGA In Clinical Products
Determination of Q1 sameness of PLGA between the test and reference products can be challenging as the composition of the PLGAs used in clinical products are not readily available and very limited studies have been conducted focusing on establishing experimental methods for determining critical characteristics of PLGA. A study was conducted to develop and validate an analytical protocol to determine some key characteristics of PLGA used in commercial microspheres, such as molecular weight, monomer ratio (L:G), and polymer end-group[16]. Trelstar® (3.75 mg strength) and Risperdal® Consta® were chosen as model products. The calculated L:G ratios of the PLGA in Trelstar® and Risperdal® were found to be 52:48 and 78:22, respectively. It was also found that the PLGA from both Trelstar® and Risperdal® possessed ester end-caps. Additional studies are currently ongoing to identify critical characteristics of PLGA for assessment of Q1 sameness.
Conclusions
These research studies have furthered the understanding of in vitro and in vivo performance of PLA/PLGA-based drug products and how in vitro studies and modeling tools could be used in generic drug development and review. OGD plans to conduct additional research studies for PLA/PLGA-based drug products as understanding of these products evolves. The outcomes from these studies may be used for developing new approaches to demonstrate bioequivalence for these products in the future.
References
- 21 CFR 314.94(a)(9)(iii)
- FDA. GDUFA Regulatory Science. Available at: http://www.fda.gov/ForIndustry/UserFees/GenericDrugUserFees/ucm370952.htm.
- J. Shen, W.Qu, S. Choi, Y. Wang, and D. Burgess, In vitro and in vivo performance testing of risperidone microspheres. American Association of Pharmaceutical Scientists, 2014.
- J. Shen, W.Qu, S. Choi, Y. Wang, and D. Burgess, Development of an in vitro release testing method for porous PLGA microspheres. American Association of Pharmaceutical Scientists, 2014.
- J. Shen, N.Tipins, S. Choi, W. Qu, Y. Wang, and D. Burgess, In vitro-in vivo correlation of risperidone microspheres. Controlled Release Society Annual Meeting, 2015.
- J. Andhariya, J.Shen, W. Qu, Y. Wang, S. Choi, and D. Burgess, Storage stability tests of risperidone-loaded PLGA microspheres. American Association of Pharmaceutical Scientists, 2015.
- J.Shen, W.Qu, Y. Wang, S. Choi, and D. Burgess, Effect of manufacturing process parameters on physicochemical properties of peptide microspheres. American Association of Pharmaceutical Scientists, 2015.
- N. Swarnakar, J.Shen, W. Qu, S. Choi, Y. Wang, and D. Burgess, Development of an accelerated in vitro release testing method for vivitrol®. American Association of Pharmaceutical Scientists, 2015.
- J. Shen, W.Qu, S. Choi, Y. Wang, and D. Burgess, Effect of manufacturing processes on in vitro performance of peptide microspheres. Controlled Release Society Annual Meeting, 2016.
- J. Andhariya, N.Swarnakar, J. Shen, W. Qu, Y. Wang, S. Choi, and D. Burgess, Effect of manufacturing processes on critical quality attributes of naltrexone microspheres. Controlled Release Society Annual Meeting, 2016.
- A. Doty, Y.Wang, S. Choi, W. Qu, R. Lionberger, M. Feng, A. Schwendeman, S. Schwendeman, Cage implant system to evaluate mechanisms of in vivo PLGA microsphere release for IVIVC model development. American Association of Pharmaceutical Scientists, 2014.
- A. Doty, K.Hirota, R. Ackermann, K. Olsen, R. Feng, W. Qu, Y. Wang, S. Choi, A. Schwendeman, and S. Schwendeman, A cage implant system for assessing in vivo controlled release performance of long-acting release PLGA microspheres. American Association of Pharmaceutical Scientists, 2015.
- K. Hirota, A.Doty, R. Ackermann, K. Olsen, M. Feng, Y. Wang, S. Choi, W. Qu, A. Schwendeman, and S. Schwendeman, Characterizing release methanisms of leuprolide-loaded PLGA microparticles for IVIVC development. American Association of Pharmaceutical Scientists, 2015.
- S. Schwendeman, K.Hirota, A. Doty, Y. Wang, S. Choi, and W. Qu, Mechanistic evaluation of in vitro and in vivo release from PLGA microspheres employing a cage model. 14th European Symposium on Controlled Drug Delivery, 2016.
- J. Shen, S.Choi, W. Qu, Y. Wang, and D. Burgess, In vitro-in vivo correlation of parenteral risperidone polymeric microspheres. Journal of Controlled Release, 2015. 218: p. 2-12.
- J. Shen, K.Lee, S. Choi, W. Qu, Y. Wang, and D. Burgess, A reproducible accelerated in vitro release testing method for PLGA microspheres. International Journal of Pharmaceutics, 2016. 498: p. 274-282.
- J. Garner, S.Skidmore, H. Park, K. Park, S. Choi, and Y. Wang, A protocol for assay of poly(lactide-co-glycolide) in clinical products. International Journal of Pharmaceutics, 2015. 495: p. 87-92.