MSD R&D (China) Co., Ltd.
MSD R&D (China) Co., Ltd.
The dry powder inhaler (DPI) device is paramount to the success of a DPI product. It is the vehicle the formulation is delivered through for local or systemic effect via pulmonary the route. The successful delivery of drugs into the deep lung depends on the integration between device performance and powder formulations. The combination of the device and the formulation needs to demonstrate safety, efficacy, bioequivalence and reliability for product approval. Many factors affect the device performance. Some of the factors include mouth piece configuration1, grid structure and mouthpiece length2, impaction angle of the powder with devices3 and air inlet size4. Due to the complexity and various configurations of the device, there are numerous device designs. However, review articles regarding DPI devices are limited5-8. This review is to give a brief summary of the currently available devices and highlights some new developments. DPI devices can be categorized as capsule based, blister based, canister/cartridge based and other types. Only one or two products in each category will be discussed due to page limits. Readers are strongly suggested to refer to the patent number of the products for detailed information.
Capsule Based Devices
Capsule based devices generally have a chamber to store a capsule. The capsule is broken by external force by action of twist or pins. Powder is released from the capsule upon aspiration. Capsule based devices are listed in Table 1.
Table 1. Summary of capsule based inhaler devices
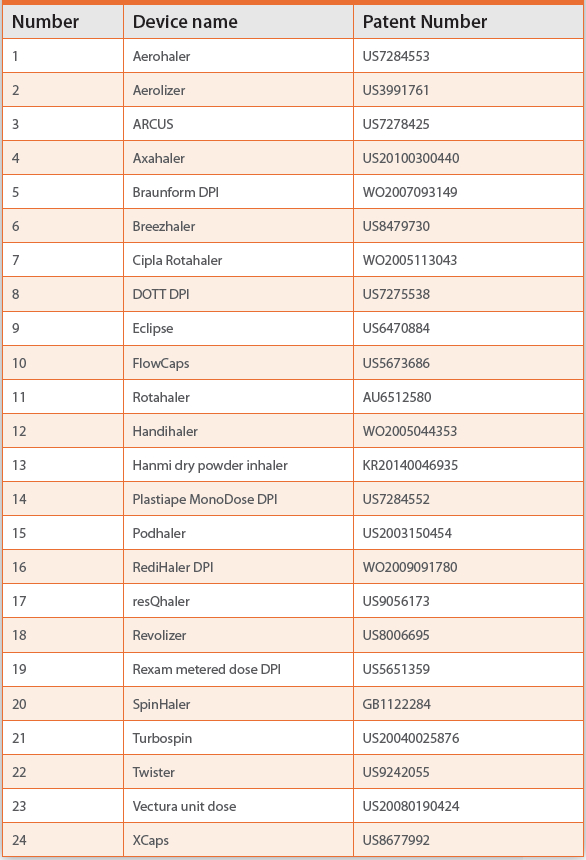
 Figure 1. Illustration of Rotahaler
Figure 1. Illustration of RotahalerA simple capsule based device is the Rotahaler as shown in Figure 1. The device has a barrel-shaped cap and body shell. The cap has two holes, one for capsule insert and the other one is for air inlet during aspiration. The body serves as a mouthpiece to be inserted into mouth. There is a grid between the cap and the body. The grid has many functions, it generates small eddies and allows high speed collisions for the drug particles. The space between cap and grid is the capsule chamber. When in use, the patient inserts the capsule prefilled with the drug into the cap. The drug powder is released from the capsule into the device chamber by twisting the cap and body. Powder will fluidize when the patient breathes through the mouthpiece. Drug particles pass through the grid and eventually deposit in the lung following the air stream.
 Figure 2. Illustration of Aerolizer. Picture taken from literature5
Figure 2. Illustration of Aerolizer. Picture taken from literature5Another capsule based device is the Aerolizer (Figure 2). It has a capsule chamber in the base of the inhaler, ports for air inlet, pins near the air inlet, grid in between the capsule chamber and the mouthpiece. The powder is released by piercing the capsule by the pins.
Blister Based Device
Blister based devices normally have a ring of aluminum blisters inside the device. Each blister contains one dose of drug pre-dispensed. The device also has a dose counter as a dosing indication. Drug powder is released by piercing the blister before inhalation. The drug powder is carried away by the air stream created by the patient’s inhalation.
An example is the Microdose DPI (Figure 3). The microdose DPI has a piezoelectric vibrator converting electrical energy to mechanical motion. A blister is pierced before the patient breathes. Activation of the piezoelectric vibrator is upon detection of a threshold level of inspiratory airflow by the patient. The Piezoelectric vibrator is used to deaggregate the drug powder packaged in aluminum blisters. More details of blister based devices can be found in Table 2.
 Figure 3. Illustration of Microdose DPI. Picture taken from literature9
Figure 3. Illustration of Microdose DPI. Picture taken from literature9Table 2. Summary of blister based inhaler devices

Reservoir/Cartridge Based Device
The reservoir/cartridge based device has a powder chamber to store medicament. The device has a mechanism to dispense the powder each time during inhalation. This Multiple use device has a dosing meter. Table 3 summarizes the available reservoir/cartridge based devices.
Table 3. Summary of reservoir/cartridge based inhaler devices

 Figure 4. Novolizer, picture taken from literature10
Figure 4. Novolizer, picture taken from literature103M Conix 1 is an example for single use reservoir based device. A powder blend is sealed in a cone shape cyclone chamber. Air is drawn into the cyclone chamber upon aspiration. The airflow reverses and travels up through the circular upon reaching the bottom of the chamber. Particles are separated by the swirling air created by the cyclone. Drug particles are then carried toward the central reverse vortex through the outlet.
The Novolizer (Figure 4) is a multiple use cartridge based device. It has a button connected with a lever. The lever is then connected with a sliding bar. A powder chamber containing the drug is located above the sliding bar. A Metered amount of drug powder is stored in a metering cavity besides the sliding bar. The metered drug powder is then moved to powder channel through the sliding bar when the button is pressed. Drug powder in the powder channel is then inhaled into the lung through the cyclone based mouthpiece.
Other Types of Inhaler Devices
The Taper DPI (US8985102) has microstructured carrier tape containing the API in small micro-depressions (“dimples”). Within the device, a fixed length of the microstructured carrier tape is presented into the dosing zone prior to deliver of a dose. The amount of API delivered with each dose is determined by the number of dimples, the volume of each dimple and the density of API powder packed into the dimples.
Another interesting device is the Respira AOS (US8651104). It has an inlet channel through which air enters the inhaler, and a chamber that receives air from the inlet channel. The chamber contains a bead-like ball. The ball is coated with drug. At the end of the chamber there is a mesh. The ball oscillates upon inhalation between the mesh and the device wall. This oscillation and impaction of the ball with obstacles will release the drug coated on the surface of the ball. The drug then passes through the mesh to the lung.
A drinking straw like tube is used in the TriVair (US5797392). A single dose of API is placed in the straw sealed by a removable cap. A patient removes the cap, inserts the tube into the mouth and inhales to release the API.
Add-Ons
Add-ons aimed at improving patient compliance have been developed over the past few years. These add on features include airflow and dose monitoring. These add-ons have sensors and Wi-fior Bluetooth connection. The airflow and dosing information can be uploaded and shared with smartphones. They help patients monitor their delivery efficiency. The information gathered can also remind the patient when it is time to take the next dose. Example devices include SmartTurbo, SmartDisk, SmartMat and Propeller.
Add-ons to improve aerosolization are also being developed. One of the examples is a cyclone connected to the end of the inhaler device. By testing with Cyclohaler, Accuhaler, and Easyhaler with the spacers the deposition of particles is improved11. Another example is a spacer between the inhaler device and the mouthpiece12. This helps deaggregate agglomerates.
Conclusion
Drug powder inhalation is a fast growing area. The device is an essential component for a successful inhalation product. This review summarizes major types of devices for dry powder inhaler. Detailed information can be found in the reference patents.
References
- Coates M, Chan H-K, Fletcher D, Chiou H 2007. Influence of Mouthpiece Geometry on the Aerosol Delivery Performance of a Dry Powder Inhaler. Pharm Res 24(8):1450-1456.
- Coates MS, Fletcher DF, Chan H-K, Raper JA 2004. Effect of design on the performance of a dry powder inhaler using computational fluid dynamics. Part 1: Grid structure and mouthpiece length. Journal of Pharmaceutical Sciences 93(11):2863-2876.
- Adi S, Tong Z, Chan H-K, Yang R, Yu A 2010. Impact angles as an alternative way to improve aerosolisation of powders for inhalation? European Journal of Pharmaceutical Sciences 41(2):320-327.
- Coates MS, Chan H-K, Fletcher DF, Raper JA Effect of design on the performance of a dry powder inhaler using computational fluid dynamics. Part 2: Air inlet size. Journal of Pharmaceutical Sciences 95(6):1382-1392.
- Chan H-K, Young PM, Traini D, Coates M 2007. Dry powder inhalers: challenges and goals for next generation therapies. Pharmaceutical technology erupoe:19-24.
- Islam N, Gladki E 2008. Dry powder inhalers (DPIs)—A review of device reliability and innovation. International Journal of Pharmaceutics 360(1–2):1-11.
- Frijlink HW, de Boer AH 2005. Trends in the technology-driven development of new inhalation devices. Drug Discovery Today: Technologies 2(1):47-57.
- Hoppentocht M, Hagedoorn P, Frijlink HW, de Boer AH 2014. Technological and practical challenges of dry powder inhalers and formulations. Advanced Drug Delivery Reviews 75:18-31.
- Brown BA-S, Rasmussen J, Becker D, Friend DR. 2004. A piezo-electronic inhaler for local & systemic applications. ed., Drug development and delivery.
- Kohler D 2004. The Novolizer®: overcoming inherent problems of dry powder inhalers. Respiratory Medicine 98, Supplement 1:S17-S21.
- Parisini I, Cheng SJ, Symons DD, Murnane D 2013. Potential of a Cyclone Prototype Spacer to Improve In Vitro Dry Powder Delivery. Pharm Res 31(5):1133-1145.
- Matida EA, Finlay WH, Rimkus M, Grgic B, Lange CF 2004. A new add-on spacer design concept for dry-powder inhalers. Journal of Aerosol Science 35(7):823-833.