One of the most frequently asked questions by the end user of endotoxin testing systems is “How can I make my endotoxin test as time efficient as possible while still running a fully compliant test?” This article offers a number of ways in which you can perform a BET compliant test with your current methodologies, while minimizing the time to successfully complete the assay.
Rapid Microbiological Methods (RMMs) are an essential tool used for continuous monitoring of the pharmaceutical manufacturing processes with higher sensitivity/precision producing reproducible test results within a much shorter time frame when compared to the conventional, growth-based microbiological methods.
The Bacterial Endotoxins Test (BET) is considered to be a RMM although listed within the US Pharmacopeia among biological methods.1 BET assays allow the detection and quantification of endotoxin as a biologically active element of Gram-negative bacterial cells which may be found within manufacturing processes even when following successful sterilization/disinfection procedures. The BET assay is routinely used for monitoring water systems, testing raw materials and in-process samples and is required for release of finished drug products and medical devices. The BET assays were introduced more than 40 years ago as an invitro alternative to the Rabbit Pyrogen Test (USP Chapter <151>).
The very first BET assay referred to as the gel-clot method, with a fixed incubation time at 1 hour, was considered to be a rapid test at that time. The launch of the turbidimetric and chromogenic methods utilizing instrumentation (e.g. absorbance readers) and software added the option of acquiring test results even faster - well under an hour.
So how do we make a rapid test even faster? The procedural steps of the BET assay can be broken down into two phases: test preparation and incubation period. The test preparation includes preparation of reagents, the setup of required controls, sample dilutions and their controls. The incubation time period, however, may be variable according to the type of method and the desired sensitivity along with other test conditions. This article lists several options to complete both phases of the routine BET assay in an efficient manner.
Test Preparation Time
The preparation time of the test at the bench can tremendously impact the workflow within the BET laboratory along with overall turnaround time.
Standard dilution series
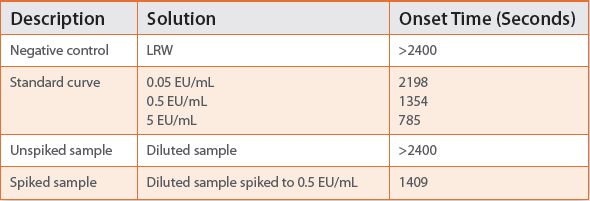
Per the BET Pharmacopeial chapters,1,2 every routine photometric test has to include negative controls, a full standard curve, sample spiked and unspiked in order to be compliant (as shown in Table 1).
Table 1.

The individual endotoxin standard concentrations for the standard curve are prepared from a standard stock solution. It is desirable to prepare as few dilutions as possible (with dilution factor of no more than 1:100) in order to minimize laboratory error. Three standard concentrations can be used for the standard curve.
For example, a kinetic chromogenic assay with the sensitivity of 0.05 EU/mL may be completed within 50 minutes with 10 minutes of preparation time and a 40 minute runtime allowing for sufficient separation of negative controls (see Table 2).
Table 2.
Automation
Automation of the liquid handling steps in laboratories with high through-put of samples can significantly reduce the preparation time, laboratory cost and error rates. There are a number of commercially available liquid handling platforms that can be configured to handle a variety of routine procedures (e.g. preparation of stock solutions, calibration curves and sample dilutions).
Archived curves
The use of archived standard curves may decrease the time needed to prepare controls.
Please note that the practice of archiving standard curves is not currently described in any of the regulatory literature. As such, it is considered to be an alternative procedure and should be validated and shown to achieve equivalent or better result than the use of calibration curves run concurrently with the samples with each assay.3-5 In addition, an archived curve is specific to the test conditions (e.g. lot combination of lysate and endotoxin standard). Any change in the test conditions requires a new archived curve.
Following the successful validation, a routine test typically contains the following:
Table 3.

By making up a single standard endotoxin concentration to spike both the diluted sample (for the spiked sample) and the spiked LRW (for positive controls), the preparation time may be decreased by approximately 10 minutes.
Incubation Time
The incubation time for the assay is dependent on several factors that are described below.
Rapid Gel-Clot Test
ACC developed Pyrosate®, as an FDA licensed gel-clot reagent that uses variable incubation times. It relies upon the same principles as the kinetic photometric tests in that the assigned sensitivity for each kit will reach its gelation point at different times with different endotoxin concentrations. Pyrosate® is available in 3 different sensitivities with variable incubation time depending on the sensitivity of the kit. For instance, for the kit sensitivity of 0.25EU/ mL, the incubation time is usually as short as 20 minutes.
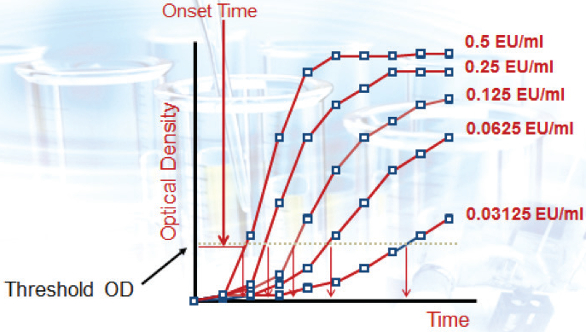 Figure 1.
Figure 1.Kinetic Photometric Methods
The principle of the kinetic photometric methods is different when compared to the gel-clot method. The typical readout for each photometric test sample is referred to as the “onset time”. Onset time is characterized as the time needed to reach a pre-set Optical Density (OD) threshold value (e.g. 20 mAbs) and is usually expressed in seconds.
When the onset time of the unknown sample is compared against the onset times of the standards, an endotoxin value is interpolated from the calibration curve.
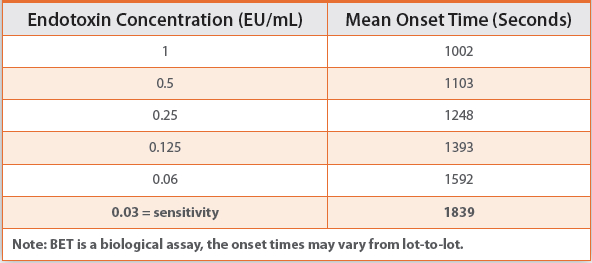
Table 4 depicts the typical speed of the kinetic turbidimetric reaction. The concentration of 0.03 EU/mL reacts in 30 minutes (instead of 60 minutes for the gel-clot). Even after allowing additional time (e.g. 5 minutes) for negative control separation, the total runtime is reduced to approximately 35 minutes achieving the same assay sensitivity as with the gel-clot test.
Table 4.
Required Test Sensitivity Versus Assay Runtime
Ultimately, as shown in Table 4, the speed of the kinetic BET reaction is inversely proportional to the chosen limit of detection (LOD). This means that the higher the LOD, the faster the test. The LOD for the kinetic photometric tests is defined as the lowest concentration on the standard curve.
Some reagents are capable of testing to a maximum sensitivity of 0.001EU/mL (with an onset time around 6000 seconds) which can be extremely beneficial when testing complex samples (needing high dilution) or samples with low endotoxin limits. However, if Wfisamples are being tested to an endotoxin limit of NMT 0.25 EU/mL, a less sensitive assay (e.g. with LOD of 0.125 EU/mL) may be used which will dramatically reduce the assay runtime (e.g. to 25 minutes as shown above for the kinetic turbidimetric test).
Please note that decreasing the test sensitivity will decrease the Maximum Valid Dilution (MVD). Ensure that the sample dilution under test does not exceed the MVD.
Lowering the OD Threshold
For the chromogenic method, decreasing the threshold value from 30 mAbs to 20 mAbs will decrease the onset time by a few minutes with no impact on the resolution of the calibration standards.
The OD threshold is a value optimized by lysate manufacturers’ for their respective reagent as described in the Instructions for Use. It should be determined what value is best for your assay and sample type. Once a finished product is validated using a particular onset OD, that value should be used consistently for routine testing.
Table 5.

Table 6.
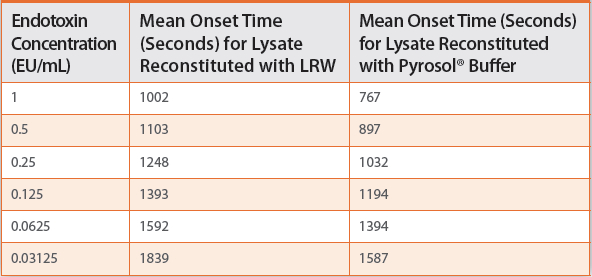
Using Buffers
The buffers used for the reconstitution of ACC’s LAL reagents provide optimal reaction conditions (pH and ionic forces) for the test samples and increase the speed of the LAL reaction. This translates into shorter onset times for the calibration standards, which in effect, shortens the assay time even further. The same turbidimetric test is now completed within 30 minutes.
Other Considerations
 Figure 2.
Figure 2.It is also important to note that each laboratory has unique requirements. A solution to make a test more “rapid” in a laboratory with a large number of samples may be very different than for a smaller laboratory. Also, a laboratory that tests predominantly water samples will have very different needs to those laboratories that test more complex samples requiring sample treatment or other types of handling.
A “rapid” test that only allows testing one sample at a time may in fact not be as “rapid” as another type of test where 20 samples can be completed simultaneously, including the setup, all within an hour.
Selecting a suitable photometric instrument is key when considering assay time. There are two instruments commonly used for running the kinetic photometric BET assays: the incubating MicroPlate Reader and the Tube Reader. The former is a universal laboratory instrument while the latter is a BET specific instrument.
 Figure 3.
Figure 3.For those laboratories looking for a rapid assay, both are capable of completing a BET compliant test in 20-30 minutes depending on the chosen assay sensitivity. The Plate Reader is a closed system that once the test is in progress, does not allow for modifications such as adding more samples or sample dilutions.
The Pyros Kinetix® Flex tube reader is an open access system, which allows the user to have continuous access and controlled addition of samples to the test while in progress as samples arrive in the laboratory. In a fast paced laboratory, this can help to significantly reduce both technician time and reagent costs.
This provides the most efficient use of time for the assay yielding sample results as they are tested.
Conclusions
In summary, there is no “one-fits-all” answer to speeding up the already rapid BET test. It is dependent on the needs of the laboratory, the expected throughput of samples, the available instrumentation and staff, the types of samples being tested, endotoxin limits along with other assay factors.
The experienced staff of Associates of Cape Cod, Inc. is always available for a free consultation to provide expertize and guidance on how to increase efficiency and turnaround time while not compromising the regulatory needs, accuracy and precision of the results.
Reminder
The information in this article is given for the purposes of education and discussion. It is not intended to be, and it should not be used, as a substitute for the regulations or regulatory guidance. Decisions and actions should be based on the relevant regulations, guidance documents and Pharmacopeial chapters, not on this article.
References
- Bacterial Endotoxins Test Chapter <85>. USP 39, United States Pharmacopeial Convention, Rockville, MD.
- Bacterial Endotoxins Test, European Pharmacopoeia 7, chapter 2.6.14, 171-175.
- FDA Guidance for Industry, Pyrogen and Endotoxins Testing: Questions and Answers, 2012
- General Notices 6.30. Alternative and Harmonized Methods and Procedures. USP 39, United States Pharmacopeial Convention, Rockville, MD.
- Validation of Compendial Procedures Chapter <1225>. USP 39, United States Pharmacopeial Convention, Rockville, MD.
Veronika Wills serves as the Associate Manager, Technical Services at Associates of Cape Cod, Inc. Her interests include biochemistry, microbiology and immunology, and she holds a Master’s Degree in Biochemical Engineering. Veronika has been with Associates of Cape Cod, Inc., for nearly 10 years and conducts BET training workshops in the U.S. and abroad.