There are many patients for whom traditional tablets and capsules are not ideal dosage forms. It is difficult to persuade infants and very young children to swallow them, and they may pose a choking hazard. Older children may simply not want to take them. Adults can have difficulties, too, for example bariatric patients often find swallowing a challenge, and those who have Parkinson’s disease can be particularly badly affected.
Orally disintegrating tablets (ODTs) provide an alternative to overcome these challenges. They resemble a traditional tablet, but have one important difference: they disintegrate rapidly in the mouth, and therefore do not need to be swallowed. As well as the patient groups identified above, a significant proportion of the general population finds swallowing tablets difficult, and an ODT can greatly increase compliance. No liquid is required when taking the medication either, which is a significant advantage when on the go.
The rapid onset of action is of benefit in drugs designed to treat acute conditions, such as migraines and psychiatric incidents, as well as conditions like insomnia. Furthermore, if the drug is absorbed within the oral cavity rather than being digested, it avoids the first pass of the liver. This pre-gastric absorption can reduce side effects caused by metabolites formed by liver enzymes.
Zydis® ODT technology was first developed by RP Scherer (now part of Catalent) in the 1980s, and the first products reached patients in the early 1990s. More than 35 products have been launched using this technology in more than 60 countries around the world. More than a billion of the ODTs are manufactured every year in Catalent’s UK facility in Swindon.
Several alternative ODT technologies are available, as can be seen in Table 1.
Table 1.
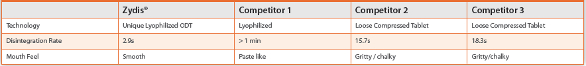
The Zydis freeze-dried oral solid dosage form typically dissolves in the mouth in under three seconds, which is significantly less than the other ODTs, and has a smooth mouthfeel. Only one of the three other methods outlined in Table 1 uses lyophilization, and tablets made this way typically take more than a minute to disperse. The other two technologies are both loosely compressed tablets, which take between 15 and 20 seconds to disperse.
Table 2 gives a list of examples of commercial products that are formulated using Zydis technology.
Table 2.

Most are relatively low doses of small molecule APIs, although doses up to 200mg have also been marketed and peptide and protein products have also been commercialized. They cover a wide gamut of therapeutic areas, with the majority in areas that benefit from the fast onset of action.
How are the ODTs made?
The basic framework of the Zydis ODT is provided by matrix and structure forming ingredients. The matrix typically comprises bovine or fish gelatin, although non-gelatin polysaccharide polymers can also be used. A structure former, usually mannitol – chosen for its high compatibility and its well-known documented properties - is also added. Additional excipients are frequently incorporated, as well as these include high-intensity sweeteners, such as aspartame, acesulfame K and sucralose, pH modifiers, flavors and colors. All of these are well established and widely accepted ingredients in pharmaceutical formulations.
To make the tablets, the excipients are first dissolved or suspended in purified water, and the API added. The solution or suspension is dosed into a pre-formed blister tray using a positive displacement pump, with the volume accurately measured to ensure the precise dose of API is present in each individual blister mold.
The filled trays are passed through a liquid nitrogen freeze channel, causing the API solution or suspension to freeze very rapidly. The trays are stored in a freezer until a sufficient quantity has been prepared to fill the freeze dryer, and then they are transferred into the dryer.
Within the freeze dryer, the shelves are heated gently under a very low pressure, causing the ice crystals present in the blister pockets to sublime. As the water is vaporized without going through the liquid state, it does not redissolve the solid ingredients, instead, leaving behind a porous gelatin framework. The trays are removed from the dryer, the blisters sealed and split into strips, and finally, packed in cartons ready for delivery to the patient.
Improved Side-Effect Profile: An Example
Pre-gastric absorption can have significant advantages in terms of both a faster onset of action and the reduction of side-effects. As an example, the drug selegeline is a monoamine oxidase B inhibitor used to treat Parkinson’s disease and depression, but its active metabolites include methamphetamine, which leads to side-effects. If the active does not pass through the liver first, these drawbacks will be reduced. A lower dose is required as the active is not metabolized by the liver before it reaches the bloodstream.
The graph in Figure 1 shows a comparison between 10mg doses of selegeline in both traditional tablet and ODT formats, and a 1.25mg Zydis ODT. The area under the curve for the low-dose ODT is equivalent to that of 10mg formulated conventionally. Furthermore, Figure 2 shows that the levels of metabolites are significantly lower with the ODT, too.
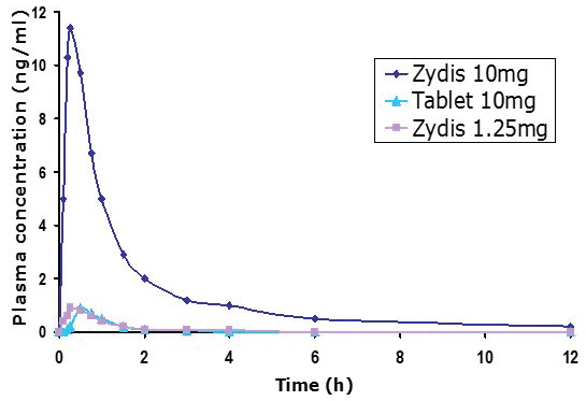 Figure 1.
Figure 1.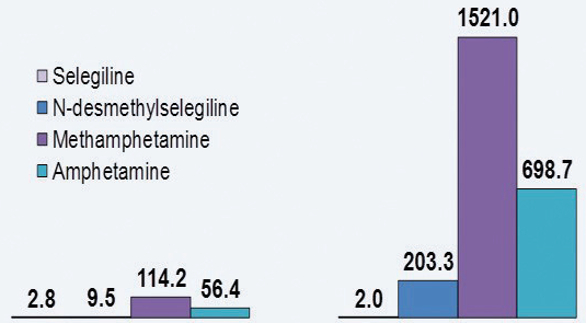 Figure 2.
Figure 2.Not all APIs can be formulated in such a way that pre-gastric absorption is possible. It is also dependent on the dose, lipophilicity and molecular weight of the drug molecule. However, if the API is suitable, then careful ODT formulation can be used to ensure the drug is absorbed in the oral cavity rather than in the gastrointestinal tract.
Importantly, an ODT formulation can be bioequivalent to a traditional dosage form. As can be seen in Figure 3, the area under curve (AUC) over time is the same for the ODT taken both with and without water, and the standard capsule formulation.
 Figure 3.
Figure 3.Next Generation ODTs
ODT technology has not remained static over the past 30 years. It is, for example, possible to make bi-layered tablets that permit two otherwise incompatible ingredients to be delivered in the same dosage form. This is achieved by first dosing and freezing the first ingredient in solution or suspension, then dosing the second ingredient (again in solution or suspension) on top and freezing again before they have time to interact with each other. The freeze drying process then proceeds as normal, followed by packaging.
By sequentially dosing two separate matrix solutions with different gelling or density characteristics in this way, a single ODT with acceptable quality characteristics can be achieved. This can either contain two different APIs, or an API and a necessary excipient that is otherwise incompatible. It is also possible to create ODTs from emulsions rather than suspensions or solutions of APIs. As an example, a proof-of-concept tablet was created using a 15% w/w emulsion of olive oil in water. Using otherwise standard ODT formulation technology, freeze dried ODTs, each containing 75mg of olive oil, were successfully made. The tablet retained all the fast dispersion characteristics of a standard ODT. This would permit very lipophilic APIs to be formulated via dissolution in oil before freeze drying.
Making ODTs More Palatable
One drawback to the dispersion of a tablet in the mouth is its taste. Many APIs have an unpleasant taste or create other unappealing sensations as they are solubilized. Careful taste-masking can greatly improve compliance by making the ODT more palatable. In many cases, if a residual taste is only moderately unpleasant for patients, including flavor ingredients and sweeteners in the formulation can be sufficient to overcome this.
However, if it is very bitter, or produces a burning or numbing sensation, more sophisticated taste-masking techniques will be necessary. One strategy involves the use of cyclodextrins. These sugar-based macromolecules, often used as odor removers in household products, have a hole in the middle, allowing smaller molecules to fit inside. If the size of the hole is matched to the API, it can trap the unpleasant tasting active, preventing it from coming into contact with the tongue’s taste receptors.
This strategy has been successfully applied to the antihistamine cetirizine, which has a very bitter taste. In a taste evaluation study of an ODT formulation incorporating beta-cyclodextrin, more than 80% of a group of 30 subjects judged the taste profile to be acceptable. The AUC was almost identical to that for a regular cetirizine tablet.
More recently, the Zydis Ultra formulation has been developed, which incorporates coated APIs for taste-masking purposes. The coating is applied via a novel, proprietary method. As Figure 4 shows, the ODT made using a standard fluidized bed coating technique is released very rapidly, whereas those using the new LabRAM ResonantAcoustic® Mixer technique have a retarded release, which is indicative of tastemasking. The USP criteria for immediate release are still met.
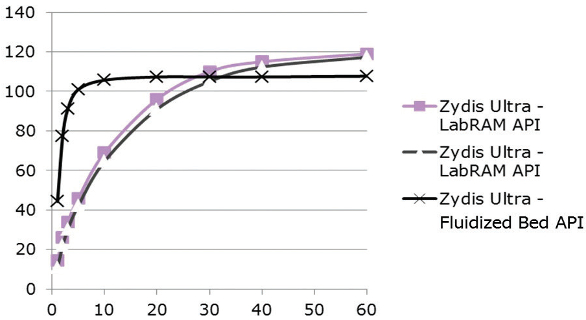 Figure 4.
Figure 4.The coating technology involves the API particles and micronized polymer agglomerates being placed in a mixing vessel equipped with an acoustic vibrator. This vibrates, causing the particles to move and accelerate by as much as 100 times the force of gravity. Initially, the polymer particles are deposited in a discrete polymer layer around the outside of the API particles, but the combination of the vibration and the impact of collision with the milling media cause these closepacked coating particles to deform into a continuous polymer layer.
In contrast to normal coating methods, no solvent is required, and the API retains 70–85% potency w/w. Particles as small as 100μm can be coated, significantly smaller than the 200–400 μm particles that conventional coating is successful for. This is important, as the larger the particles, the more gritty and unpleasant the mouthfeel will be in an ODT. Both ibuprofen and acetaminophen have been successfully formulated in this way to date.
Towards Oral Biologics
Biologic medicines are rarely orally available, not least because they cannot withstand the harsh acidic and enzyme-rich conditions in the gastrointestinal tract. It also dilutes the dose, and the high molecular weight can lead to absorption problems. By administering these molecules sublingually or buccally, these problems might be avoided.
The inclusion of bioadhesives and absorption enhancers can promote absorption. Indeed, the site of absorption, the mouth, gives no extreme pH exposure as salivary pH is typically about 6.8, and no relevant proteases are present.
Catalent’s recently developed Zydis Bio is able to present active macromolecules in a robust, convenient and fast-dispersing form. An additional advantage is the potential to avoid the need for cold-chain storage. The lyophilization based manufacturing process and its low temperatures reduces the potential for heat damage to the biologics. Careful selection of formulation options and ingredients, including the selection of the matrix component and the pH, can optimize inprocess stability.
The ODTs have the potential for good long term stability, not least because of the low water activity in the dried product. The aluminum packaging provides a high moisture barrier and good environmental protection, and acceptable storage has been demonstrated at room temperature for multiple peptide and protein compounds.
As an example, the 32 amino acid peptide calcitonin, which has a molecular weight of 3432, was formulated as an ODT. Figure 5 shows the difference between a standard ODT and the Bio ODT. The Bio formulation, administered sublingually, gives a significantly higher AUC.
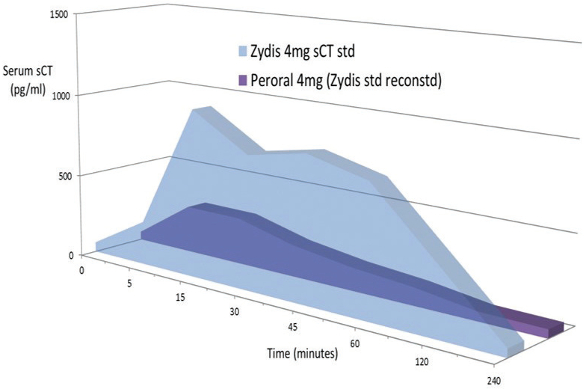 Figure 5.
Figure 5.The Bio ODT also holds out promise for oral vaccines. This would almost certainly improve compliance, and offer an improved safety profile compared to an injectable vaccine. Furthermore, it could help to generate a mucosal response, which is important for certain infections such as influenza, pneumonia and human papillomavirus (HPV). The convenience of the dosing, and the lack of requirement for cold chain storage, could permit faster distribution in pandemic and emergency situations, and would permit administration by less qualified healthcare professionals. This is important for use in a developing world setting.
Development work has been carried out using influenza as a model vaccine antigen. In the graph in Figure 6, the vaccine was administered sublingually to mice before they were challenged with influenza virus. Body weight loss, which is an indicator of the extent of the disease, was greatest for the unvaccinated and infected control group, which is demonstrated by the results shown on the lowest set of data points on the graph and shows rapid weight loss after day 2 of the study. In the most effective vaccine formulation, there was very little change in body weight across the period of monitoring.
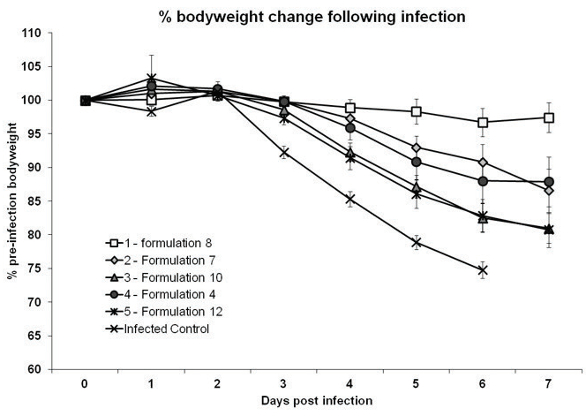 Figure 6.
Figure 6.Although it is still early days for ODT formulation of biologics, the model studies already carried out indicate its clear potential. In the long run, if such formulations gain regulatory acceptance, they could significantly improve the delivery of both biologic medicines and vaccines, particularly in environments where a lack of clean water and trained nurses present challenges to traditional drug administration.