The milling of drug substances to reduce particle size is a common process used in the pharmaceutical industry. Reducing particle size helps achieve uniform distribution of the drug when blended with excipients, which is especially important for low dose products (drug content < 5% w/w). Reducing particle size also increases the available surface area of a given mass of drug substance and consequently increases the rate of dissolution in a solvent. The relationship between the rate of dissolution and solubility of a drug substance is described by the Noyes-Witney equation (Figure 1) which shows that the rate of dissolution is directly proportional to the surface area of the solid.
 Figure 1. Noyes-Whitney equation.
Figure 1. Noyes-Whitney equation.‘micronisation’. The typical particle size range achievable by jet milling is 2 – 50 μm.
Micronization remains a useful approach for improving dissolution and bioavailability of drug substances but it has limitations where aqueous drug solubility is particularly low and also for high doses. The production of sub-micron particles, nanoparticles, can significantly increase the dissolution rate of poorly water-soluble drug substances compared with micronized material, simply as a consequence of increasing the total surface area of the resulting particles. A fascinating illustration of the effect of particle size reduction on increased surface area is given as follows: if a 2 cm3 cube of material (length of each side ~ 1.25 cm) is serially divided twenty four times a single layer of the resulting 1 nm sized cubes would completely cover the surface of a football pitch!
The proportion of poorly water-soluble compounds emerging from drug discovery pipelines is increasing year-on-year. In fact, the majority of new small molecule drug candidates now fall into this category. Consequently, pharmaceutical scientists use a range of formulation technologies (e.g. drug nanosizing, self-emulsifying systems, amorphous solid dispersions) to encourage drug dissolution in aqueous media and hence aid drug absorption. But choosing which of these technologies is the most appropriate for a given compound is not often obvious.
The Developability Classification System (DCS)1 has provided a useful framework for selecting a formulation technology for a drug substance based on an assessment of its fundamental properties and dose expectations. The DCS is represented in Figure 2. It allows classification of a drug substance according to the ratio of intended dose to aqueous solubility, and its predicted intestinal permeability.
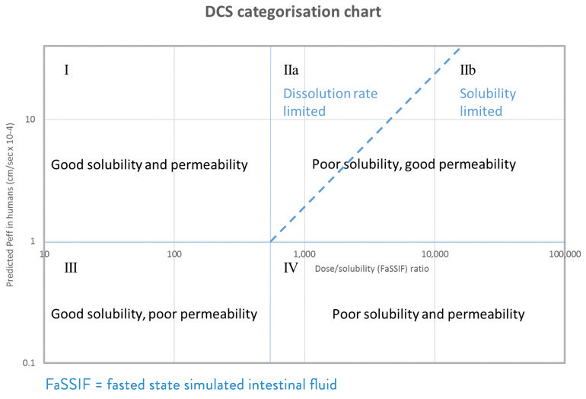 Figure 2. Developability Classification System (DCS).
Figure 2. Developability Classification System (DCS).Of particular importance is the the subdivision of Class II compounds (which make up the majority of poorly water-soluble drugs) into those for which absorption of the required dose can be considered dissolution rate limited (IIa) and those for which absorption of the required dose is solubility rate limited (IIb). For category IIa compounds it should be possible to achieve complete absorption of the dose by means of particle size reduction using conventional processing (including micronization). For category IIb compounds a drug solubility enhancing approach is generally required to achieve satisfactory absoption of the required dose.
Figure 3 shows the distribution of different technologies used for a wide selection of drugs. Interestingly, drug nanoparticle presentations have been used effectively for compounds falling just within the IIb category (elliptical area), even though strictly speaking this is a particle size reduction/dissolution enhancement approach. The reason for this behavior is the increased saturation solubility as well as the rapid dissolution rate which can be exhibited for nanosized particles.
 Figure 3. Distribution of formulation technologies used for different categories of drugs.
Figure 3. Distribution of formulation technologies used for different categories of drugs.There are several means by which nanosized drug particles can be produced, which are broadly split into particle size reduction (topdown) or controlled particle precipitation (bottom-up) processes.
Top-down technologies are by far the most often used for producing nanosized drug particles. Wet bead milling consists of the attrition of drug particles in suspension with milling media (metallic or ceramic beads) as described by Merisko-Liversidge and Liversidge2 and illustrated in Figure 4.
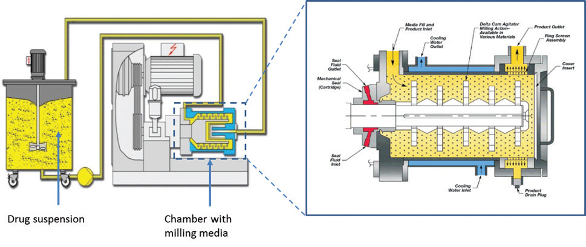 Figure 4. Wet bead milling process for producing nanosuspensions.
Figure 4. Wet bead milling process for producing nanosuspensions.An alternative technology used for producing nanosuspensions is highpressure homogenization, which involves forcing a drug suspension through a small orifice under high pressure. In this process particle size reduction is achieved through cavitation. Both these technologies are capable of producing nanoparticles in the range of 100 – 500 nm. The use of vibratory milling3,4 to produce nanosuspensions also shows great promise. The technology uses low-frequency, high-energy sound waves to mix/agitate powders or suspensions within a container. By incorporating milling media (e.g. ceramic beads) in the suspension it is possible to produce sub-micron particles of a drug substance through the intense attrition caused by the movement of the beads within the suspension. An example of the degree of particles size reduction achievable by nanomilling and the effect on drug dissolution is illustrated in Figure 5.
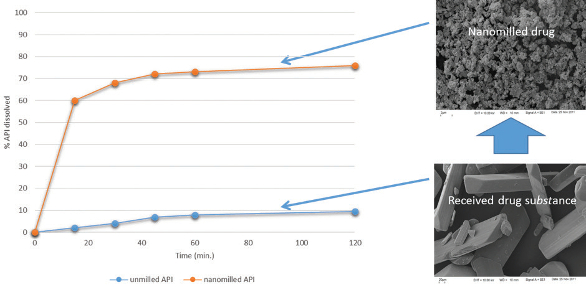 Figure 5. Effect of nanomilling of fenofibrate on dissolution rate (FaSSIF medium).
Figure 5. Effect of nanomilling of fenofibrate on dissolution rate (FaSSIF medium).The bottom-up approach5 for producing nanosuspensions involves pre-dissolving the drug in a suitable solvent and then changing the environment to cause precipitation of the drug. Precipitation may be induced through removal of the solvent (e.g. spray-drying or freezedrying) or by introduction of an anti-solvent.
For all techniques described above a crucially important element to the formulation is the use of an effective stabilizer system. A stabilizer system is required to prevent the formation of non-dispersible aggregates due to the electrostatic attractive forces between nanoparticles and to prevent particle size growth through Ostwald ripening of nanoparticles. Typically, the stabilizers which are used are either particle surface modifiers (e.g. ionic and non-ionic surfactants) or hydro-colloids (e.g. hydroxypropylmethylcellulose (HPMC) and often both types of stabilizer are used in the same formulation. The way in which these stabilizers work is by coating the surface of the nanoparticles, thereby providing a physical barrier between particles, thus preventing particles from getting close enough to aggregate.
Nanosuspensions alone have limited applicability for oral administration, with most commercial oral dosage forms being powder/ granule-based capsule or tablet presentations. There is therefore usually a necessity to convert a nanosuspension intermediate into a more conventional oral dosage form through additional downstream processing.
There are a variety of methods which can be used to convert a nanosuspension into a solid form. The most popular processes are fluid-bed granulation and spray-drying. For the fluid-bed granulation approach a carrier material (e.g. lactose) is fluidized in an air stream and the carrier particles spray-coated with the nanosuspension. The solvent (water) is removed during this process resulting in a layer of nanoparticles over the surface of each carrier particle (see Figure 6). An alternative approach is to dissolve a water-soluble carrier excipient (e.g. mannitol) in the aqueous nanosuspension and to spray dry the resulting combined nanosuspension/carrier solution. The carrier excipient precipitates as the water is driven off during processing leaving a coating of the drug nanoparticles embedded on the surface of the spray-dried carrier particles (see Figure 7). In this dried form the coated particles can easily be incorporated into formulations for convenient processing into capsules or tablets.
 Figure 6. Lactose particles coated with fenofibrate nanoparticles (using fluid bed granulation).
Figure 6. Lactose particles coated with fenofibrate nanoparticles (using fluid bed granulation). Figure 7. Mannitol particles coated with fenofibrate nanoparticles (using spray-drying).
Figure 7. Mannitol particles coated with fenofibrate nanoparticles (using spray-drying).The technologies for developing and processing drug nanoparticulate dosage forms are now well established, having been used by the pharmaceutical industry for over twenty years. Many products incorporating drug nanoparticles have been developed and licensed globally, a selection of which is shown in Figure 8.
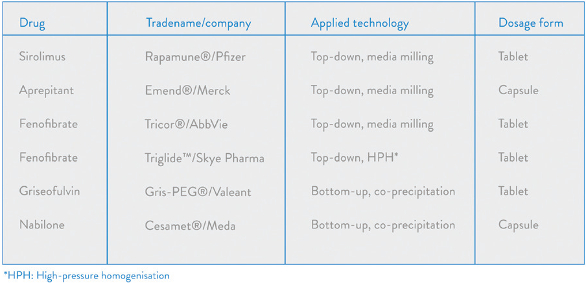 Figure 8. Current marketed oral products based on nanoparticulate technologies.
Figure 8. Current marketed oral products based on nanoparticulate technologies.With the increasing number of poorly water-soluble drugs emerging from drug discovery it is inevitable that the use of dissolution and solubility enhancing technologies will continue to be play an important role in drug product development. Drug nanosizing technologies remain an attractive option when enhanced drug dissolution is necessary. Both top-down and bottom-up technologies are becoming more established in pharmaceutical manufacture with many examples of commercial products available worldwide. And with the introduction of new, more economically efficient processing technologies for producing nanosuspensions, such as vibratory milling, we can expect the number of products based on the use of drug nanoparticulate platforms to increase in the coming years.
References
- Butler, J.M. and Dressman, J.B. The Developability Classification System: Application of biopharmaceutics concepts to formulation development. J. Pharm. Sci. (2010), 99: 4940- 4954.
- Merisko-Liversidge, E. and Liversidge, G.G. nanosizing for oral and parenteral drug delivery: a perspective on formulating poorly-water soluble compounds using wet media milling technology. Adv. Drug Deliv. Rev. (2011), 63: 427-440.
- Leung, D.H., et al. A new and improved method for the preparation of drug nanosuspension formulations using acoustic mixing technology. Int. J. Pharm.(2014), 473: 10-19.
- Li, M., et al. An intensified vibratory milling process for enhancing the breakage kinetics during the preparation of drug nanosuspensions. AAPS PharmSciTech (2016), 17(2): 389- 399.
- Du, J., et al. nanosuspensions of poorly water-soluble drugs prepared by bottom-up technologies. Int. J. Pharm. (2015), 495:738-749.