Summary
A study was conducted to investigate the effect of headspace pressure within the prefilled syringe system where liquid leakage (dripping) was observed from the syringe needle upon removal of needle shield. Drip tests were performed to measure the expelled quantity on syringes manually filled with 0.5 or 1.0 mL of various aqueous solutions. Parameters evaluated included temperature (filling and test), bulk storage conditions (tank pressure and gas type), and user test procedures (syringe orientation). Results showed that leakage was indeed driven by increased in headspace pressure, but the temperature effect (ideal gas expansion) was not the only factor. In addition to the expected ideal gas expansion, soluble gas was found to be a key factor causing increased air headspace pressure. Overall, leakage amount was observed to be a function of bulk storage pressure, gas solubility, filling temperature, and needle-shield removal procedure. Results suggested that the internal air headspace pressure of a prefilled syringe can be minimized by reducing bulk storage pressure, reducing filtration pressures, filling at a higher temperature, or by employing lower solubility gas, such as Helium, for bulk transfer and storage. Pressure effects could also be mitigated by holding the syringe needle upward during needle shield removal.
Introduction
Prefilled syringe has been the preferred primary container for many parenteral drug delivery systems because they are convenient and have lower risk of contamination compared to vials [1]. However, there are factors, as subtle as how the needle shield is removed, that may affect dosing accuracy. We recently found that a small amount of solution (up to 3-4% of the 1mL filled volume) leaked from a needle upon removal of the needle shield / tip cap from a syringe held vertically with the needle pointing downwards. Although holding the syringe tip downwards during needle shield removal may not be a standard technique, this issue could significantly affect the overall delivery dose, especially in small fill volumes.
 Figure 1. Process flow diagram of syringe filling operation.
Figure 1. Process flow diagram of syringe filling operation.During the filling process at a production scale, bulks are often pressurized up to 10 psig with an inert gas, such as Nitrogen, for an extended period of time in a storage bulk prior to fill (see Figure 1). Thus it was hypothesized that the presence of dissolved gas during filling could be a contributor to the elevated headspace pressure. In addition, syringes may be filled from cooled bulk solutions, so gas expansion caused by the temperature differences between storage/fill (5 to 15°C) and usage (ambient) is expected. Gas pressure increasing upon warming may be due to both the effects predicted by the Ideal Gas Law, and also to the decrease in gas solubility accompanying higher temperature. The solubility of gases, Nitrogen in this case, in an aqueous phase, is a function of temperature, pressure, solution composition, and gas species. In general, the solubility of gases in aqueous solutions increases with increased pressure and decreases with increased temperature. Therefore, it is important to understand the behavior of dissolved gases and control the manufacturing and filling process parameters in order to minimize the internal pressure in all aqueous-based protein solutions.
This study investigated whether and how the temperature and pressure history of the liquid may eventually affect headspace pressure. Furthermore, procedures of needle shield removal were assessed to better understand the leakage phenomenon.
 Figure 2. Apparatus of drip test experiment.
Figure 2. Apparatus of drip test experiment.Materials and Methods
All experiments were carried out using 1.0 mL long 26G ½ inch staked needle syringes, filled with 1.0mL or 0.5mL volume, containing pure water or an anti-IgE monoclonal antibody aqueous solution. All fluids were filtered with 0.22μm PVDF membrane and stored at 2-8°C under a pre-determined pressure prior to filling. Syringes were manually filled under the laboratory environment. Samples were analyzed by measuring gross weight change using an analytical balance before and after removal of the rigid needle shield at ambient condition (see Figure 2).
The following sections describe the processing history of solutions used in this experiment and procedures of the syringe filling operation.
Liquid Bulk Storage Under Pressurization
A 5 L stainless steel pressure vessel was assembled with a calibrated pressure gauge, a valve connected with a vent filter, and a pressure regulator connected with gas source. Each filtered solution was individually poured into the pressure tank and pressurized to the desired pressure (5, 10, or 20 psig) and held at 2-8°C for 24 hours. The pressure gauge was checked periodically to ensure constant pressure was maintained.
Sterilizing Components
Empty syringes with 26G ½ inch staked needle, (previously washed and siliconized by an external contract manufacturer), W4023/ FLT stoppers, and stopper insertion tools were autoclaved using a cycle with conservative ramp rates during pressurization and evacuation to minimize the chance of needle shield movement during the sterilizing process. After the cycle was complete, each syringe was visually inspected for loose needle shield.
Manual Syringe Filling and Stoppering
The pressure vessel was depressurized to 0 psig by closing the gas source and opening the vent filter valve. A repeater pipette was used to withdraw solution from the vessel and filled into 1 ml-long syringes, with fill volumes of 1.0 mL or 0.5 mL. Plunger stoppers were manually inserted using a custom stopper insertion tool. The depths of the stopper (measured from the back of the syringe flange to the back of the stopper) were positioned at 8 mm for 1 mL syringes and 24 mm for 0.5 mL syringes. This resulted in headspace height of approximately 8 mm for both configurations, which represents worst case scenario at some full scale productions. Although most syringe samples were filled in the cold room at 2-8°C, some were filled at higher temperature (15°C and 21.5°C) using a circulation bath to control liquid temperatures. All filled syringes were stored in the 2-8°C cold room for at least 24 hours to allow the headspace pressure to equilibrate.
Drip Test Method
Unless specified, all filled syringes were equilibrated to ambient temperature for at least 30 minutes before testing. The initial weight of the syringe sample, Winitial, was measured using the digital balance. Syringes were then held vertically with the needle pointing downward (except for samples tested for different RNS removal methods), and the needle shield was removed to allow any solution to drip from the tip of the needle onto a water-absorbent material. When dripping stopped, the weights of the syringe and needle shield, Wafterdrip, were recorded. Finally, to determine the overall fill volume, a plunger rod was inserted to inject all liquid content from the syringe. Empty syringe weight, Wempty, was then determined, and the gross weight of dripped solution was calculated accordingly.
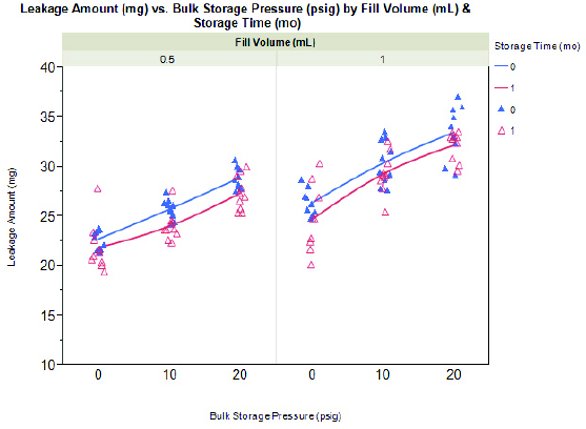 Figure 3. Effect of liquid bulk storage pressure (0, 10, and 20 psig) on leakage from syringes filled with 1.0mL or 0.5mL of anti-IgE antibody liquid formulation. Syringes tested at t=0 and t=1 month are represented by color blue solid triangle and pink hollow triangle, respectively.
Figure 3. Effect of liquid bulk storage pressure (0, 10, and 20 psig) on leakage from syringes filled with 1.0mL or 0.5mL of anti-IgE antibody liquid formulation. Syringes tested at t=0 and t=1 month are represented by color blue solid triangle and pink hollow triangle, respectively.Gas Expansion Calculation (Ideal Gas Law)
Based on the Ideal Gas Law (Equation 1), filling syringes at 5°C (T1) under ambient pressure (P1) and relocating them to ambient environment at use (P2 and T2), for testing would cause gas expansion in the air headspace; hence changes in volume is expected. As the rigid needle shield is being removed, the open system allowed changes in air headspace volume due to pressure differentials. This change can promote displacement of solution until the headspace pressure equilibrates to ambient conditions (P1=P2). Bubble expansion (V2) displaced some solution as leakage and the expanded volume (V2 – V1) could be theoretically calculated using Equation 2.
 (1)
(1)
 (2)
(2)
where T1 and T2 are known values, initial and final temperatures, respectively; and V1 is a constant that can be calculated from known syringe physical parameters (Equation 3).
 (3)
(3)
where BH is the barrel height, LH before drip is the liquid height before removal of the needle shield, S height is the stopper total height, S Depth is the stopper position, measured from the back of the syringe flange to the back of the stopper, and BCA is the barrel cross-sectional area.
Results and Discussion
Effect of Bulk Storage Pressure and Temperature
Drip tests were first performed on syringes hand-filled with the anti- IgE antibody formulation that have been stored under Nitrogen blank at three different pressures: 0 psig, 10 psig, and 20 psig. Results are summarized in Figure 3. The ideal-gas expansion of inert Nitrogen illustrated was calculated using Equation 2 with the temperature change between filling and drip test environments, from 5°C to 21.5°C, respectively. When the needle shields were removed, both 0.5 mL and 1.0 mL configurations showed increase in drip volumes as the storage pressures increased, approximately 0.3 mg/psig. The 1.0 mL samples appeared to expel more liquid, 25-33 mg (corresponding to 6-8 drips or 2.4%-2.8% of the overall dose) compared with 23- 29 mg (corresponding to 5-7 drips or 4.6%-5.0% of the overall dose) for the 0.5 mL samples over the tested pressure range. These observations could be attributed to the presence of soluble gas. When bulk storage pressure increased, more gas molecules were dissolved into the solution. As 1.0 mL filled syringes contained twice the liquid volume compared to 0.5 mL filled syringes, more dissolved gas diffused from the liquid to the gaseous phase when syringes reached equilibrium temperature at ambient.
 Figure 4. Effect of gas species (Helium vs. Nitrogen) on leakage from syringes filled with 1.0mL or 0.5mL of anti-IgE antibody liquid formulation. Syringes with fill volumes of 1.0mL and 0.5mL are illustrated by pink hollow square and blue hollow circle, respectively
Figure 4. Effect of gas species (Helium vs. Nitrogen) on leakage from syringes filled with 1.0mL or 0.5mL of anti-IgE antibody liquid formulation. Syringes with fill volumes of 1.0mL and 0.5mL are illustrated by pink hollow square and blue hollow circle, respectivelyNote that most values were observed to be higher than the Ideal Gas Law prediction from gas expansion, even with 0 psig storage condition. This was believed to be contributed by the initial dissolved gas existing in the 0 psig solution during filling at 2-8°C and became insoluble and would diffuse to the headspace, which caused higher pressure and additional leakage.
Additional syringes tested after 1 month storage at ambient showed similar trend to previous observations, where displaced liquid volumes increased with increasing storage pressures for both 0.5 mL and 1.0 mL samples.
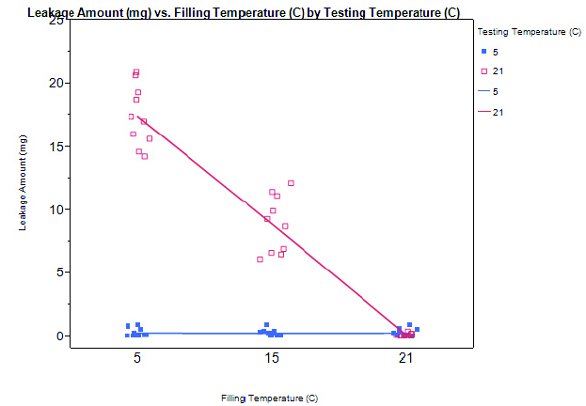 Figure 5. Effect of filling temperature (5°C, 15°C, and 21.5°C) and drip test temperature (5° [blue solid square] and 21°C [pink hollow square) on leakage from syringes filled with 1.0mL of pure water.
Figure 5. Effect of filling temperature (5°C, 15°C, and 21.5°C) and drip test temperature (5° [blue solid square] and 21°C [pink hollow square) on leakage from syringes filled with 1.0mL of pure water.Effects of Gas Species
Gas solubility is known to be a function of temperature, pressure, and interaction between gaseous and liquid phases [2,3]. As mentioned in the previous section, soluble gas such as Nitrogen may have contributed to headspace pressure increase; a less soluble gas may alleviate such an effect. Helium is less soluble compared to Nitrogen, 0.7 x 10-5 mole fraction, and is independent of temperature changes. Drip tests were performed on syringes filled from bulks that were again pressurized with three different pressures using Helium: 0, 10, and 20 psig. As bulk storage pressures increased, both 0.5 mL and 1.0 mL samples showed no changes in drip amount (see Figure 4). Again, the 0 psig results exceeded what was predicted by the Ideal Gas Law and can be explained by the existence of soluble gas, containing oxygen and Nitrogen from air, that already dissolved in the solution during storage and filling.
Effect of Temperature Alone (Liquid Filling and Drip Test)
Temperature appeared to be the most critical factor influencing the amount of solution leakage, particularly distinguishable on the temperature differential between filling and test / usage. Assuming the liquid was filled and tested under the same ambient pressure, the leakage amount with samples tested at higher temperature showed a negative relationship as the filling temperature increases; that is, the higher the filling temperature, the lower the leakage amount. This was related to the positive temperature differential between filling and test, causing the headspace to expand. In contrast, when the drip test was performed inside the cold room, the temperature differential was essentially zero or negative, depending on the filling temperature, hence no leakage was observed with headspace contraction. This result (see Figure 5) confirmed that leakage can be minimized or completed avoided if the liquid fill temperature is identical or higher than the drip test temperature. Thus, performing liquid fills at higher temperatures (if protein stability allows) would be an effective approach to mitigate leakage when the needle shield is removed.
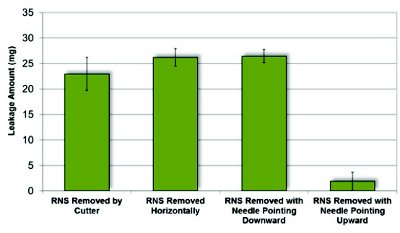 Figure 6. Effect of rigid needle shield removal methods on leakage from syringes containing 1.0mL of pure water fi lled at 5°C and 0 psig.
Figure 6. Effect of rigid needle shield removal methods on leakage from syringes containing 1.0mL of pure water fi lled at 5°C and 0 psig.Effect of Needle Shield Removal Method
Finally, needle shield removal procedures were evaluated for the impact on liquid leakage. A subset of syringes manually filled with 1.0 mL pure water (under 0 psig bulk storage pressure) were tested by holding the syringe either vertically (needle tip pointing up and down), horizontally, or by carefully cutting the needle shield open (lengthwise into halves) to minimize any vacuum that may be created from pulling on the rubber part of the needle shield. As shown in Figure 6, the vertical and horizontal removal methods showed no significant differences and the amount of leakage remained high (25-27 mg). The cutting removal method had a mild effect in reducing leakage (20-25 mg), suggesting leakage contributed by vacuum from removing the needle shield was minor. Vertically removing the needle shield with the needle pointing upwards was determined to be the most effective in minimizing leakage (2-5 mg). As the syringe was being inverted, the air headspace flowed to the top of the syringe and permitted air to vent or relief through the needle. This appeared to be the simplest solution to mitigate solution leakage, but enforcing such instruction to end users could be challenging.
Conclusion
From this study, we demonstrated that the root cause of leakage was dictated mainly by the temperature and pressure history of the liquid bulk during the filling process. Headspace pressure increase was primarily caused by Ideal Gas expansion and soluble gas re-equilibration between the liquid and the gas phases. In order to minimize the issue of leakage, the following actions can be considered:
- Minimize the pressure used for the bulk filtration process and bulk storage.
- Replace Nitrogen gas with a lower soluble gas, e.g. Helium, in bulk processing and storage.
- Fill syringes at higher temperatures, when permitted by protein stability properties, to reduce gas expansion due to temperature variation.
References
- Berends, W. Development of a Prefilled Syringe: Filling of Pre-filled Syringes. Presented at PDA Europe Conference “The Universe of Pre-Filled Syringes & Injection Devices,” Venice, Italy, 2009.
- Smith, N.; Kelemen, S.; Nagy, B. Solubility of natural gases in aqueous salt solutions, Geochimica et Cosmochimica Acta 1961, 24 (1–2), 23–31.
- Gevantman, L. H. Solubility of Selected Gases in Water. In CRC Handbook, 86th ed.; CRC Press LLC: Boca Raton, Florida, 2004, pp 8-86 – 8-87.
Author Biographies
Edwin Chan is a process engineer at Genentech under Pharmaceutical Processing & Technology Development. He is primarily responsible for developing prefilled syringes to support the company’s pipeline. Edwin received his Bachelor of Science degree in chemical engineering from University of California, Berkeley and is currently pursuing his MBA at Santa Clara University.
Yuh-Fun Maa is a Principal Engineer at Genentech focusing on prefilled syringe process development. He has over 20-years industrial experience at Genentech, Johnson & Johnson, PowderJect, and Zosano Pharma. He holds a Ph.D. in Chemical Engineering from the University of Rochester (Rochester, NY) and has authored more than 50 peer-reviewed scientific articles and patents/patent applications.
David Overcashier is a Principal Engineer in Device Development at Genentech. His work focuses on prefilled syringe technology and test-method development, and implementation for protein drug candidates. David received his BS degree from the University of California, Davis, and his MS from the University of Washington, Seattle – both in Chemical Engineering.