Introduction
Drug efficacy and bioavailability are largely dependent on drug physicochemical properties such as water solubility, pKa and Log P. Today it is widely recognized that up to 60% of new drugs in the developmental stage are water insoluble [1] and this presents a major challenge to the pharmaceutical industry. For drugs to be administered intravenously, drugs formulated as aqueous-based injections are most desirable. Thus drug aqueous solubility is a prerequisite [2]. For oral drug delivery, drugs must be able to dissolve in the aqueous gastrointestinal fluid before absorption can take place in the gastrointestinal tract (GIT) [3]. Therefore, for drugs which have high permeability but with poor solubility (Class II according to Biopharmaceutics Classification System), rate of dissolution is a major rate limiting step for the absorption of these drugs via oral administration. As a result, this undesirable property often leads to poor oral bioavailability and erratic GIT absorption.
Conventional excipients such as surfactants, cosolvents are often added to improve drug solubility for both oral and parenteral routes. Complexation with cyclodextrins or formulating oil in water emulsions are also employed to improve poorly soluble drugs. Today the growing demand to fabricate drug delivery systems capable of encapsulating high levels of active ingredients with additional benefits such as drug targeting capability has resulted in a plethora of technologies exploiting the unique physical properties of nano-sized structures. Nanotechnologies such as nano-crystals, solid lipid nanoparticles (SLN), nano-emulsion, polymeric nanoparticles, liposomes and polymeric self-assemblies have been employed to enhance drug solubilization. In this review, we will focus on four types; namely nano-crystals, SLN, liposomes and polymeric self-assemblies.
Nano-crystals
Nano-crystals technology essentially involves a nano-sizing method, in which large insoluble drug crystals are milled to form nano-sized particles with less than 2000nm as defined in the first patents in this field (Table 1) [4]. To prevent particle aggregation, excipients such as surface active agents and polymers are normally required for stabilization [5]. The decrease in drug particle size to nanoscopic crystals results in an increased surface area to volume ratio [4] (Fig 1). According to Noyes Whitney equation, the dissolution rate is proportional to the surface area of the drug particles in contact with the dissolution medium [3]:
 is the rate of dissolution of a solid, Cs is the saturation solubility of the dissolution layer, D is the diffusion coefficient of the dissolved solute, σ is the thickness of the diffusion layer and A is the surface area of the solid.
is the rate of dissolution of a solid, Cs is the saturation solubility of the dissolution layer, D is the diffusion coefficient of the dissolved solute, σ is the thickness of the diffusion layer and A is the surface area of the solid.
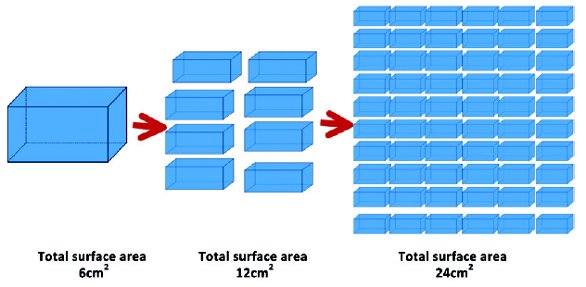 Figure 1. Surface Area Enlargement by Particle Size Reduction [4]
Figure 1. Surface Area Enlargement by Particle Size Reduction [4]Therefore, the increased surface area of nano-crystals will consequently result in an enhanced dissolution rate of hydrophobic drugs. This leads to an improvement in oral bioavailability and rate of oral absorption. Additionally, this technology can also be employed for intravenous administration where nano-crystals can be formulated as nano-suspensions for injections. Today, there are a number of products utilizing this technology that have been approved by FDA (Table 2).
Solid Lipid Nanoparticles (SLN)
Solid lipid nanoparticles (SLN) have been investigated since the early nineties as one of nano-sized drug delivery systems for hydrophobic drugs (Table 1). These particles typically have a mean particle size of 50-1000nm and composed of three essential components (solid lipids, surfactants and water) [6,7]. Different types of solid lipids such as glycerides, waxes, and fatty acids have been used in the fabrication of SLN. Depending on the fabrication method, the type, concentration of the drug, surfactant and lipid, poorly soluble drugs can be molecularly dispersed within the lipid matrix or formed a core-shell model (drug-enriched shell or drug enriched core)[6]. The advantages of using SLN include the versatility in using these delivery systems for various routes of administrations such as oral, parenteral, dermal, ocular and the manipulation of different solid lipids and production parameters to achieve optimal drug loading and release profiles. However, there are limitations such as poor loading capacity and risk of instability during storage.
Table 1. Types of Nanotechnologies used to Improve Drug Solubility

Liposomes
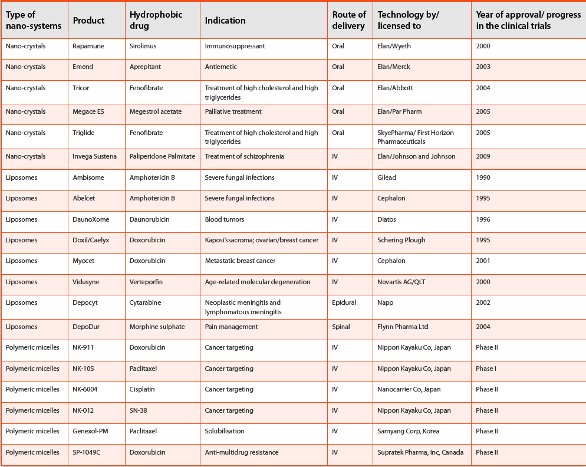
Liposomes are spherical structures composed of a phospholipid bilayer surrounding an aqueous reservoir [8]. Liposome vesicles are composed of unilamellar or multilamellar lipid bilayers which have alternate aqueous layers sandwiched between the bilayers [9] (Table 1). Upon aggregation, multilayered liposomes are formed but are easily converted to unilamellar entities by sonication [10]. In general hydrophilic compounds are not well entrapped in aqueous core of liposomes compared to hydrophobic compounds which are well retained within the phospholipid bilayers [9, 11]. Their major disadvantage as drug carriers is their instability upon drug entrapment [12]. The inclusion of drug into the lipid bilayer causes penetration and results in premature drug release [12, 13]. To combat this problem, cholesterol is incorporated into the phospholipid bilayer, consequently the liposome possess higher rigidity leading to more stable vesicles [14, 15]. Furthermore, poor stability is experienced on storage in aqueous phase. However, lyophilization and cryoprotection of the liposomal formulation have been reported to minimize this problem [12, 16]. Liposomes have been shown to improve the therapeutic efficacy of pharmaceutical drugs including ibuprofen, amphotericin B and doxorubicin [12,17,18]. Today, liposomes are one of the successful nano-delivery systems that have entered the clinics. There are currently 12 liposomal products in the market, a number of which undergo various phases of clinical trials (Table 2) [19]. The approved products are mainly administered intravenously for cancer therapy or treatment of systemic fungal infection (Table 2).
Table 2. Types of Nanotechnology Products Approved on the Market/ in Clinical Trials. (This list is not exhaustive. Please refer to [4, 19, 30] )
Polymeric self-assemblies
Polymeric self-assemblies have been widely investigated since the early 1990s as promising delivery systems for hydrophobic drugs [20]. They are formed spontaneously by amphiphilic polymers which consist of hydrophobic and hydrophilic segments. In aqueous environments, a hydrophobic core is formed spontaneously upon aggregation of the hydrophobic moieties while the hydrophilic segments will remain in contact with the aqueous environment (Fig 2). Hydrophobic drugs are encapsulated in the core via hydrophobic interactions and thus solubilization increases in aqueous environments [21]. The most common type of self-assemblies formed by amphiphilic polymers are polymeric micelles [20, 21], although they have also been reported to form a wide range of nano-structures such as vesicles and nanoparticles [22]. Amphiphilic polymers can be fabricated from diverse architectures, such as block copolymers (the most common type) [23, 24], hydrophobically modified polymers [25, 26], star shaped polymers [27] and dendrimers [28]. Unlike micelles formed by traditional surfactants, the polymeric micelles typically have significantly lower critical micellar concentration (CMC) (1x10-6M) compared to traditional micelles. As a result, they do not lose their payload upon dilution in vivo. This unique characteristic has been utilized for delivering a wide range of hydrophobic drugs such as paclitaxel, propofol, doxorubicin, etoposide and camptothecin via intravenous administration. Recently,these structures have also shown to increase oral absorption of class II drugs in vivo [26]. To date, a number of polymeric micelles have entered clinical trials (Table 2), although none have reached the clinic yet due to the relative young age of this technology. However, this technology is thought to advance further with the promising preclinical and clinical data demonstrated to date.
Drug targeting and Controlled Drug release Capabilities
Unlike conventional excipients which generally only enhance the solubilization of hydrophobic drugs, the use of nano-carriers has additional benefits of achieving site specific delivery especially for cancer therapy. This capability is demonstrated by intravenous administration of liposomes and polymeric micelles, where patients typically experience lower side effects compared to conventional formulations [29, 30]. The reason being the nano-size of these carriers allow them to passively target tumors through the phenomenon known as the enhanced permeability and retention effect (EPR). The blood vessels supplying the blood to cancerous tissue are leaky and disorganized. The tumor tissues also experience poor lymphatic drainage. Nano-structures (<120nm) are capable of entering the highly permeable blood capillaries which supply the rapidly growing tumors [29]. This however does not occur in normal tissue as the blood vessels are well formed and non-porous. Once inside the capillaries, they accumulate and are retained in the tumor as a result of the poor lymphatic drainage. Additionally, the versatility of nano-carriers enables the attachment of ligands for targeted delivery, either for cancer therapy [31] or oral delivery to promote uptake by intestinal cells [32]. Newer generations of these nano-carriers have been designed to precisely control the release of the drugs based on stimuli release mechanism. A range of external and internal stimuli such as temperature, magnetic, pH, reduced environment have been utilized to control the release of drugs demonstrating the versatility of these nanocarriers [33].
Conclusions and Future Outlook
As shown in Table 2, liposomes were the earliest nanotechnology products approved in late 1990s after the discovery in 1975 while the history of other nanotechnologies is relatively shorter than liposomes. Within the next ten years it is expected that many clinical trials using nano-sized formulations will be completed and their use in the clinics should improve treatments and patient’s quality of life. The development of biocompatible polymers/ materials for the production of nano-carriers, accompanied with advanced techniques to study the cellular uptake mechanisms and the fate of nanocarriers will fuel greater exploitation of nanotechnology in drug delivery. In the future, these new excipients could be part of the decision tree in the development and Formulation of new or existing chemical entities for enhancing solubilization of these drugs.
Author Biographies
Dr. Woei Ping Cheng joined the School of Pharmacy at University of Hertfordshire, UK as a senior lecturer in pharmaceutics in January 2009. Prior to obtaining her Ph.D. in 2005, she was a lecturer at the School of Pharmacy and Life Sciences, The Robert Gordon University, UK since December 2004. She obtained her first class BSc (Hons) Pharmacy and Ph.D. from University of Strathclyde. Her research interest is in the use of self-assembling polymers for the delivery of challenging therapeutic agents such as hydrophobic drugs, proteins and siRNA. She is the editorial board member of Drug Delivery Letters and president of UK-Ireland Controlled Release Society (UKICRS). She has been invited to chair and speak in a number of national and international scientific conferences and has published more than 35 peer-reviewed papers and conference abstracts, 3 patents and one book chapter.
Dr. Clare Hoskins joined the School of Pharmacy in Keele University, UK as a lecturer in pharmaceutics in 2011. Here she teaches on the MPharm course and continues her research in nanomedicines. Clare completed her Ph.D. in pharmaceutics at the Robert Gordon University in Aberdeen, UK in 2009. She then worked as a postdoctoral researcher at the Institute of Medical Science and Technology at the University of Dundee, UK. Here she investigated the potential use of magnetic nanoparticles for nerve regeneration in the peripheral nervous system. Now her work focuses on the investigation into hybrid metallic nanoparticles as multifunctional vehicles for imaging and targeted drug delivery for pancreatic cancer. Clare is a member of both the Royal Society of Chemistry and the UK and Ireland Controlled Release Society. Clare has been invited to peer review submissions to high quality journals as well as publishing 20 articles in peer reviewed papers and conference abstracts, along with 1 patent and 1 book chapter.
References
- Cheng.W.P, Gray.A.I, Tetley.L, Hang.T.L.B, Schätzlein.A.G, Uchegbu.I.F, 2006, Polyelectrolyte Nanoparticles with High Drug Loading Enhance the Oral Uptake of Hydrophobic Compounds, Biomacromolecules, 7, 1509-1520
- Yeh M-K, Chang L-C, Chiou A H-J, 2009, Improving tenoxicam solubility and bioavailability by cosolvent system, American Association of Pharmaceutical Sciences, 10, 166-171.
- Aulton M E, 2007 Pharmaceutics: The design and manufacture of medicines, 3rd edition, Churchill Livingstone, London UK, 2007
- Gulsun, T. Gursoy, R. Neslihan, Oner, Levent 2009 Nanocrystal Technology for Oral Delivery of Poorly soluble Drugs, FARAD Journal of Pharmaceutical Sciences, 34, 55-65
- Peltonen, L, Hirvonen, J 2010 Pharmaceutical nanocrystals by nanomilling: critical process parameters, particle fracturing and stabilisation methods. Journal of Pharmacy and Pharmacology, 62, 1569-1579
- Rainer H.M, Karsten, M., Sven G. 2000 Solid Lipid Nanoparticles (SLN) for controlled drug deliverya review of the state of the art. European Journal of Pharmaceutics and Biopharmaceutics 50, 161-177
- Souto, E.B. and R.H. Muller, Solid Lipid Nanoparticles and Nanostructures Lipid Carriers - Lipid Nanoparticles for Medicals and Pharmaceuticals. Encyclopedia of Nanoscience and Nanotechnology, 2011. 23: p. 313-328.
- Gregoriadis G, 1973, Drug Entrapment in Liposomes, Federation of European Biochemical Societies Letters, 36, 292-296
- Sinko P J, 2005 Martin’s Physical Pharmacy and Pharmaceutical Sciences 5th Edition, Philadelphia, Lippincott Williams & Wilkins
- Florence TA, Atwood D, 2006 Physiochemical Principles of Pharmacy 4th Edition, London, Pharmaceutical Press
- Nii T, Ishii F, 2005, Encapsulation efficiency of water-soluble and insoluble drugs in liposomes prepared by the microencapsulation vesicle method, International Journal of Pharmaceutics, 298, 198-205
- Mohammed A R, Coombes A G A, Perrie Y, 2007, Amino acids as cryoprotectants fro liposomal delivery systems, European Journal of Pharmaceutical Sciences, 30, 406-413
- du Plessis J, Ramachandran C, Weiner N, Muller G, 1996, The influence of lipid composition and lamellarity of liposomes on the physical stability of liposomes upon storage, International Journal of Pharmaceutics, 127, 273-278
- Kirby D J, Rosenkrands I, Agger E M, Anderson P, Coombes A G A, Perrie Y, 2008, Liposomes act as stronger sub-unit vaccine adjuvants when compared to microspheres, Journal of Drug Targeting, 16, 543-554
- Kersten G F A, Crommelin D J A, 1995, Liposomes and ISCOMS as vaccine formulations, Biochemica et Biophysica Acta (BBS), 1241, 117
- Mohammed A R, Bramwell V W, Coombes A G A, Perrie Y, 2006, Lyophilisation and sterilisation of liposomal vaccines to produce stable and sterile products, Methods, 40, 30-38
- McNeil S E, Perrie Y, 2006, Gene delivery using cationic liposomes, Expert Opinion in Therapeutic Patents, 16, 1371-1382
- Mohammed A R, Weston N, Coombes A G A, Fitzgerald M, Perrie Y, 2004, Liposome formulation of poorly water soluble drugs: optimisation of drug loading and SEM analysis of stability, International Journal of Pharmaceutics, 285, 23-34
- Chang, HI, Yeh, MK 2012 Clinical Development of Liposome-based drugs: formulation, characterisation, and therapeutic efficacy, International Journal of Nanomedicine 7,49-60
- Harada A, Kataoka K, 2006, Supramolecular assemblies of block copolymers in aqueous media as nanocarriers relevant to biological applications, Progress in Polymer Science, 31, 949-982
- Letchford K, Burt H, 2007, A Review of the formation and classification of amphiphilic block copolymer nanoparticulate structures: micelles, nanospheres, nanocapsules and polymersomes, European Journal of Pharmaceutics and Biopharmaceutics, 65, 259-269
- Thompson C, Ding C, Qu X, Yang Z, Uchegby I F, Tetley L, Cheng W P, 2008, The effect of polymer architecture on the nano self-assemblies based on novel comb-shaped amphiphilic poly(allylamine), Colloid Polymer Science, 286, 1511-1526
- Qui L, Zheng C, Jin Y, Zhu K, 2007, Polymeric micelles as nanocarriers for drug delivery, Expert Opinion in Therapeutic Patents, 17, 819-30
- Torchilin V P, 2006, Multifunctional nanocarriers, Advanced Drug Delivery Reviews, 58, 1532- 1555
- Hoskins, C., Kong Thoo Lin, P., Cheng, WP. 2012 A review on comb-shaped amphiphilic polymers for hydrophobic drug solubilisation. Therapeutic Delivery. 3, 59-79
- Hoskins C, Kong Thoo Lin P, Tetley L, Cheng WP, 2012, The use of nano polymeric self-assemblies based on novel amphiphilic polymers for oral hydrophobic drug solubilisation, Pharmaceutical Research, 29, 782-794
- Ooya T, Lee J, Park K, 2003, Effects of ethylene glycol-based graft, star-shaped, and dendritic polymers on solubilisation and controlled release of paclitaxel, Journal of Controlled Release, 93, 121-127
- Morgan M, Nakanishi Y, Kroll D J, Griset A P, Carnahan M A, Wathier M, Oberlies N U, Manikumar G, Wani M C, Grinstaff M W, 2006, Dendrimer-Encapsulated Camptothecincs: Increased Solubility, Cellular Uptake, and Cellular Retention Affords Enhanced Anticancer Activity In Vitro, Cancer Research, 66, 11913-11921
- Barenholz, Y 2012 Doxil- The first FDA approved nano-drug: lesson learned. Journal of Controlled Release 160, 117-134
- Yokoyama, M 2010 Polymeric Micelles as a new drug carrier system and their required considerations for clinical trials. Expert Opinion in Drug Delivery,7, 145-158
- Mikhail A S, Allen C, 2009, Block copolymer micelles for delivery of cancer therapy: Transport at the whole body, tissue and cellular levels, Journal of Controlled Release, 138, 214-223
- Plapied, L, Duhem, N., des Rieux A., Preat V, 2011, Fate of polymeric nanocarriers for oral drug delivery. Current Opinion in Colloid and Interface Science 16, 228-237
- Rapoport N, 2007, Physical stimuli-responsive polymeric micelles for anti-cancer drug delivery, Progress in Polymer Science, 32, 962-990