Abstract
Water uptake by a drug substance is an inherent property but also subject to its surrounding environment. During pharmaceutical development, knowing the hygroscopic nature of a drug substance is essential to the understanding of its physiochemical behaviors such as microbiological activity and stability. For setting an appropriate water specification at early development stages, this information is especially useful when limited real-time stability data is available; and quite often, water specification was set without knowing the water sorption limit. In this article, a dynamic vapor sorption (DVS) method is introduced, in which the maximum water sorption at 25°C was determined by the mass difference between 90% and 0% relative humidity (RH). Total 15 lots of two drug substances were tested by this DVS method to acquire water sorption limits. The water contents of the 15 lots under ambient temperature and humidity and accelerated storage conditions were determined by Karl Fischer (KF) titration. The results collected by the two methods were compared. The outcome of this investigation clearly demonstrates that DVS can be used to quantitatively assess the hygroscopicity, and thus, can serve as the justification of proper water specification setting. In addition, 4 lots of the same drug substance known of very difference particle size distributions were analyzed by DVS . As expected, the results support the general understanding that particle size/surface area and pore diameter played a crucial role in dictating the water sorption and desorption process.
Introduction
Water, or so-called ‘moisture’ is commonly found in pharmaceutical products. Although it is not treated as an impurity, when perceived to be a problem, water in a drug substance should be monitored and controlled as strictly as possible. USP general chapter <1241>, Water-solid Interactions in Pharmaceutical Systems, describes water-solid insteractions as adsorption (at the surface) and absorption (penetrating the bulk solid structure). The term ‘sorption’ is often used when both adsoption and absorption occur. It has been shown that the degree of water sorption affects crystallinity and influences storage modulus, permeability, density and melting point of pharmaceutical products. For amorphous material, the presence of water can significantly alter the bulk properties, for example, the glass transition temperature, and even initiate reversion to the crystalline form. Water also facilitates hydrolysis and induces drug degradation. Water activity, aW, has a direct implication for microbiological safety. In pharmaceutical industry, water content is routinely used in determining the dry and solvent-free assay value of a drug substance.
There are a number of analytical techniques that can be used for water determination in drug substances such as KF titration, loss-on-dry, and thermogravimetric analysis (TGA). KF titration listed as a compendial method has been the default technique for the water content measurement [1]. Loss-ondry and TGA are generally non-selective and limited since both volitale solvents and moisture can contribute to the weight loss, and the drug substance has to be stable at elevated temperatures. Near-infrared reflectance spectroscopy (NIRS) has recently evolved as a promising alternative technology for moisture determination as it is sensitive, rapid, non-destructive, and no sample pretreatment [2,3]. Since water sorption is the result of both the hygroscopicity of a compound and its surrounding environment, the aforementioned methods can only uncover the water content of the temperature and humidity at a specific time point, but not the potential, i.e., the maximum water sorption capacity under extreme conditions such as 90% RH. It is very obvious that without knowing the limit of water sorption, setting water specification for stability studies, especially those under accelerated conditions, can be difficult and hard to justify.
DVS, on the other hand, is capable of measuring mass changes in a controlled environment with excellent accuracy. Moisture sorption isotherms obtained by DVS analysis are one of the simplest means to study the interaction of water molecules with drug substances. DVS has been widely used in studying sorption and desorption process to explore hydrate and solvate formations [4,5] and to determine amorphous contents of a pharmaceutical material [6,7]. At Amgen, by using DVS, the maximum water uptake by a drug substance is measured by the mass difference between 90%RH and 0%RH at 25°C. This value can then serve as a basis of setting specification for water content.
Experimental
The water sorption-desorption isotherms were obtained using a DVS system capable of resolving 0.1 μg. The accuracy of the system is ± 0.5% for the RH over a range of 0-98% and ± 0.3ºC for 25ºC. At 25ºC, two RH cycles were performed for each sample. In each cycle, RH was raised in 10 steps, 10% ramp per step, from 0% to 90% and then back to 0%. A rate of change in mass per time unit (dm/dt) of 0.0005%/min was set as the equilibration parameter. At each stage, DVS will hold the set parameters for 10 minutes once dm/dt reaches 0.0005%/min. The DVS water sorption limits were calculated by averaging the mass differences between 90% RH and 0% RH of each cycle. Figure 1 depicts the representative DVS Change in Mass Plot and the water sorption measurement.

Figure 1 - Representative DVS Change in Mass Plot and Water Sorption Limit Measurement
Results and Discussion
Adsorption Mechanism and Hygroscopicity
Table 1 - Water Contents by KF Titration vs. Water Sorptions by DVS of Drug Substance A

Drug substance A has been used for clinical studies with oncology indications. Its lots 1 to 13 were produced in chronological order between 2002 and 2006 by an Amgen contract manufacturer. Table 1 lists their water sorption limits measured by DVS and compared with the water content results obtained by KF titration during release testing. As shown in Table 1, for each and every lot, the water content does not exceed the water sorption potentials indicated by DVS since the ambient storage humidity is usually below 90% RH and above 0% RH. In 12 out of the 13 lots, the maximum water sorption at 90% RH was less than 0.3%, which demonstrates that drug substance A is not hygroscopic and is well correlated with a surface adsorption mechanism. In Figure 2, the representative DVS isotherm plot of drug substance A shows there is virtually no hysteresis for each cycle considering the overall small scale of change in mass. Little to no bulk absorption was observed, most likely it was a completely reversible adsorption mechanism.

Figure 2 - Representative DVS Isotherm Plot
Table 1 - Water Contents by KF Titration vs. Water Sorptions by DVS of Drug Substance B
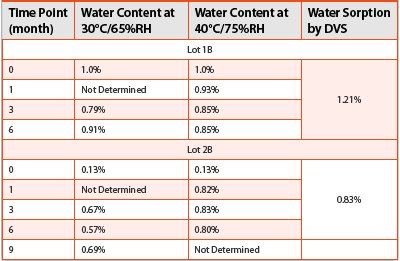
In comparison, the isotherms of two lots of a hygroscopic drug substance B were acquired by DVS following the same procedure. KF water contents were also obtained from the same two lots stored up to 9 months under conditions of 30°C/65% RH and 40°C/75% RH. The results of the maximum water sorptions of 90%RH by DVS and water contents by KF titration at each time point were summarized in Table 2. Table 2 clearly shows, for drug substance B, even at the elevated temperature and humidity, the KF results did not exceed the water sorption limits determined by DVS.
Particle Size and Water Sorption
By examining the particle size distributions and water sorption results obtained from DVS analysis in Table 1, it is noticed that the lots with smaller particle size had higher water sorption values, for example, lot 6 (D50 = 12 μm) had a water sorption of 0.23%; and lots with larger particle sizes had lower water sorption with the exception of lot 1, for example, lot 2 (D50 = 19 μm) had a water sorption of 0.14%. To further evaluate this correlation, the same DVS procedure was performed on three air-classified lots and one micronized lot with very different particle size distributions. The airclassification is a process aided by air flow to filter and segregate a drug substance by particle size. A clear trend was identified as shown in Figure 3 that the DVS water sorption values decrease as the particle sizes increase for the 3 air-classified and 2 unprocessed lots. This is in the agreement with surface adsorption of water: smaller particles will have a larger surface area onto which water may adsorb. However, the micronized lot 5 is found as an exception to this trend. Compared with the unprocessed lot 5 (D50 = 15 μm), micronized lot 5 has a smaller particle size (D50 = 9 μm), but their DVS water sorption values are equal (0.18%). This most likely is due to the physical characteristic changes during the micronization, such as bulk density and shape of particles which may alter the dynamic of water sorption and desorption processes.
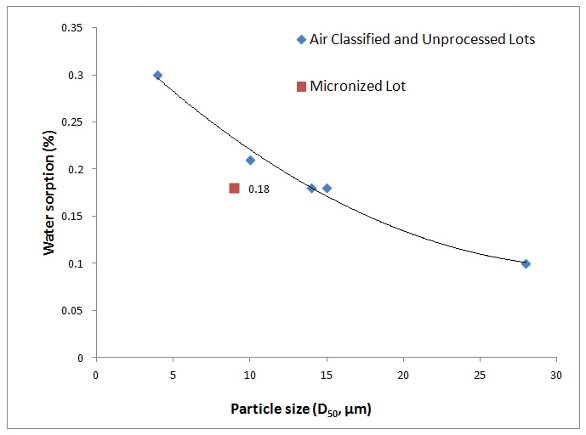
Figure 3 - Correlation of Particle Size and Water Sorption
Pore Diameter, Surface Area and Water Sorption
Table 1 - Surface Area, Pore Diameter and Water Sorption
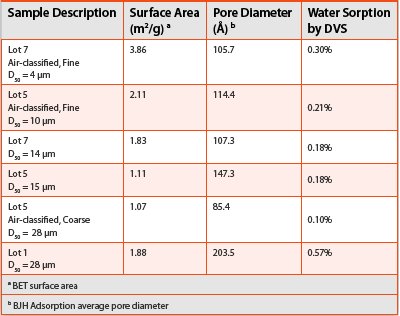
For drug substance A, both coarse lot 5 and lot 1 has the largest particle size (D50 = 28 μm, Table 3), However, lot 1 gave the highest DVS water sorption (0.57%) and coarse lot 5 gave the lowest DVS water sorption (0.10%). The DVS result of lot 1 contradicted the expectation that the larger particle size, the smaller surface area, the less surface water sorption. To demonstrate that particle size is not the only factor that influences the water sorption and desorption process of drug substance A, surface areas and pore diameters were measured by a contract test lab and compared in Table 3. For air-classified lots 7 and 5 (fine and coarse), the DVS water sorptions decrease along with the ascending particle sizes and dropping surface areas. For lot 1, its surface area is similar to that of lot 7; however, its pore diameter is almost doubled. It seems that the pronounced pore diameter, the so called ‘sponginess’ resulted in the higher water sorption of lot 1. In addition, compared with lot 7, lot 5 has a larger pore diameter, approximately 147 Å, however, its surface area is smaller, approximately 1.1 m2/g relative to the 1.8 m2/g of lot 7. Thus, the combined factors contributed to the identical DVS water sorption for lot 5 and lot 7. In summary, the results in Table 3 well represent the releationship between surface area, pore diameter and water sorption. Given the surface areas and pore diameters, it is no surprise that lot 1 has the highest water sorption and coarse lot 5 has the lowest.
DVS Repeatability Assessment
To demonstrate the precision of the DVS method, triplicate DVS determinations were performed on lot 1 of the highest water sorption and on the coarse lot 5 of the lowest water sorption. A RSD of 3.0% was achieved for the high end measurements and 7.0% for the low end measurements. The averages of three measurements, 0.58% water sorption of lot 1 and 0.11% of coarse lot 5, are very close to the 0.57% shown in Table 1 and 0.10% in Table 3. These results demonstrate that the water sorption limit measurement by the procedure described in this article is precise and reproducible.
Conclusion
The DVS results show that drug substance A is not hygroscopic. When the humidity is ramped from 0% to 90%RH at 25°C, the majority of DVS water sorption of 13 lots of drug substance A range from 0.14% to 0.23%. Physical characteristics of the material, such as particle size, surface area, and pore diameter dictate the water sorption and desorption, and their impacts can be either positive or negative. When the surface area dominates, the smaller particle size (larger surface area), the more water sorption. For all drug substances studied under various storage conditions, the water contents by KF titration do not exceed the water sorption by DVS. Thus, the water sorption results by DVS can be used as a reference to set water limit in the drug substance specification; and DVS becomes a valuable tool when setting storage conditions and handling hygroscopic materials.
Acknowledgements
The author would like to thank Ruthie Ji, Xichen Zhang, and Judy Ostovic of Amgen for their support. Special thanks to Hong-Xun Guo for his review of the manuscript.
References
- Zhou, L., Socha, J.M., Vogt, F.G., Chen, S., and Kord, A.S. “A Systematic Method Development Strategy for Water Determinations in Drug Substance Using Karl Fischer Titrations” (2010) Am. Pharm. Rev. January/February 74-84.
- Zhou, X., Hines, P., and Borer, MW. “Moisture Determination in Hygroscopic Drug Substances by Near Infrared Spectroscopy” (1998) J. Pharm. Biomed. Anal. 17, 219-225.
- Zhou, G.X., Ge, Z., Dorwart, J., Izzo, B., Kukura, J., Bicker, G., and Wyvratt, J. “Determination and Differentiation of Surface and Bound Water in Drug Substance by Near Infrared Spectroscopy” (2003) J. Pharm. Sci. 92, 1058-1065.
- Giron, D., Goldbronn, C., Mutz, M., Pfeffer, S., and Piechon, P. “Solid State Characterizations of Pharmaceutical Hydrates” (2002) J. Therm. Anal. Calorim. 68, 453-465.
- Burnett, D.J., Thielmann, F., and Sokoloski, T.D. “Investigating Carbamazepine-Acetone Solvate Formation via Dynamic Gravimetric Vapor Sorption” (2007) J. Therm. Anal. Calorim. 89, 693-698.
- Young, P.M., Chiou, H., Tee T., Traini, D., Chan, H., Thielmann F., and Brunett, D. “The Use of Organic Vapor Sorption to Determine Low Levels of Amorphous Content in Processed Pharmaceutical Powders” (2007) Drug Dev. Ind. Pharm. 33, 91-97.
- Hogan, S.E., and Buckton, G. “The Application of Near Infrared Spectroscopy and Dynamic Vapor Sorption to Quantify Low Amorphous Contents of Crystalline Lactose” (2001) Pharm. Res. 18, 112-116.
Author Biography
Jingshun Sun is a Senior Scientist at Amgen in the Analytical R&D department. He received his Ph.D. from University of Florida in 1998 and has been with Amgen since 2003. He has worked at various positions within pharmaceutical industry over 12 years. His recent work has been focused on new and novel separation and detection techniques of small and large molecules with interests spanning from chromatography to spectroscopy. Dr. Sun is a diligent and frequent author on the subjects of advocating modern analytical technologies and innovative applications.
This article was printed in the September/October 2011 issue of American Pharmaceutical Review - Volume 14, Issue 6. Copyright rests with the publisher. For more information about American Pharmaceutical Review and to read similar articles, visit www.americanpharmaceuticalreview.com and subscribe for free.