Introduction
Excipients are included in medicinal products to facilitate manufacture, consumption or administration, or to enhance stability/absorption. They can also facilitate product differentiation, enhance aesthetic appearance, and improve compliance. They are generally considered as inert viz not possessing intrinsic biological activity [1]. Antimicrobial preservatives might be considered exceptions to such categorization, being added to help improve antimicrobial stability and hence requiring antimicrobial activity. Their presence is mandated for multidose liquid and semi solid products and performance standards are defined in compendial monographs [2,3]. Compliance with such requirements is not easy. This review, being Part One of a three-part review discusses the biological mode of action of preservatives, options for selection, and the requirements for preservative efficacy testing.
Preservative Availability
Table 1 - Common Preservatives for Pharmaceutical Products

Preservatives are widely used in cosmetic and food products. A more limited number are used in medicinal products, if monographs in the major pharmacopoeias are the yardstick [4,5,6]. Preservatives comprise a range of chemical classes as illustrated in Table 1.
The listings in Table 1 illustrate the limitations available to the pharmaceutical formulator. Availability for specific products or routes of administration is further constrained by the requirements listed in Table 3. Furthermore, there are no preservatives possessing sufficient efficacy, safety and non-irritancy to allow inclusion in medications instilled into ocular or intrathecal tissue [3]. Such products must be preservative-free.
Other agents have also been used as preservatives, and can still be found in mature products [2]. These include many of the older quaternary ammonium compounds (QACs), such as cetrimide, cetalkonium chloride, cethexonium bromide, as well as hexachlorophene, ethyl alcohol, sodium metabisulphite and sulphur dioxide. The phenols, although generally discouraged in oral and to a lesser extent in topical products still have a major role to play in preserving parenteral products, particularly biologicals [10]. Similar comments apply to the organo mercurials, although their use in vaccines is still subject to ongoing controversy. The use of formaldehyde-donors e.g. imidurea and bronopol has decreased in topical and ophthalmic products due to concerns about formaldehyde sensitization. Hexachlorophene is an excellent disinfectant, but its use as a preservative has declined because of concerns over neurotoxicity [8]. Typically, the use of these older preservatives in new products has been largely discontinued because of safety considerations.
Concentrated sucrose solutions can be effective preservatives in oral liquids because of the high osmotic pressure they exert. However, sucrose has fallen from favor as a preservative or sweetener in liquid oral medications (concerns regarding dental caries) despite its almost ubiquitous presence in food and confectionary products. Propylene glycol is often used as an adjunct to other preservatives because of its good solvent properties (superior to glycerin).
Modes of Action of Preservatives
Preservatives generally offer limited protection against viral contamination. Bactericides and fungicides may evince their effects on a variety of microbial cellular targets, for example; the cell wall, the cytoplasmic membrane or the cytoplasm. It is often difficult to assign a precise target for a specifi c class of preservative; the target can and does change with preservative concentration. As a consequence, preservatives can often interfere with several different microbial cellular mechanisms (Table 2).
Table 2 - Site of Preservative Activity in Microbial Cell

Such cytotoxicity may also affect mammalian cells. Hence inclusion levels should be minimal, consistent with adequate preservation. There is a regulatory expectation that the reason for preservative inclusion, proof of efficacy, safety information, control methods in fi nished product and details of labeling in the fi nished product should all be addressed by the applicant [11]. Mechanisms for activity at the locations listed in Table 2 can also differ with each preservative as discussed below:
Cell wall activity may involve lysis due to enzyme inhibition, as is the case with phenols and organo mercurials. In contrast glutaraldehyde evinces its effect by irreversible cross-linking at the cell wall [2, 3].
Cytoplasmic membrane activity may be due to effects on membrane potential, membrane enzymatic function or general membrane permeability [2, 3]. Cetrimide, chlorhexidine, hexachlorophene, 2-phenoxyethanol, parabens and phenols affect membrane permeability allowing ‘leaking’ of essential cell constituents leading to cell death. Sorbic acid inhibits transport mechanisms across the cytoplasmic membrane and suppresses fumarate oxidation [3]. Chlorhexidine also inhibits membrane ATPase, thereby inhibiting cellular anaerobic activity. At higher concentrations it induces precipitation of cytoplasmic nucleic acids and related proteins. Other biguanides induce phase separation and the formation of domains in the phospholipid bi-layer. Chelators such as edetic acid (EDTA) compromise the integrity of the cytoplasmic membrane by chelating Ca2+ and Mg2+, making these ions unavailable to the microbial cell and potentiating other anti-microbial agents, e.g. 4-chloroxylenol [12]. Quaternary ammonium compounds bind strongly to the cytoplasmic membrane evoking general cytoplasmic membrane damage (and subsequent leakage), but particularly targeting the phospholipid bi-layer.
Cytoplasmic activity may concern uncoupling of oxidative and phosphorylation processes or interference with active transport mechanisms, as is the case with weak carboxylic acid and alcoholic preservatives. Other preservatives can inhibit electron transport chains, thereby inhibiting metabolic activity in aerobic bacteria [13]. Benzoic acid and the parabens inhibit folic acid synthesis [3]. Bronopol and other organo-mercurials target thiol enzymes [3] in the cytoplasm (as do silver compounds); whereas, formaldehyde donators e.g. imidurea act on the carboxylic and amino enzymes in the cytoplasm. Phenols cause protein denaturation [12], as do the alcohols [3].
There is the potential to select specifi c preservatives to address a particularly troublesome organism associated with a manufacturing site or process; however, eradication of these organisms is the remit of GMP (good manufacturing practice) and the preservative system should not be used to address defi ciencies in manufacturing processes. There are also possibilities for synergistic combinations to provide the requisite spectrum of activity in a particular system or product. However, the selection constraints that are discussed in Part 2 of this review, present other barriers to use of combinations of preservatives.
Choosing a Preservative
In concept, the preservative system protects the product against microbial proliferation but does not compromise product performance. In practice, this means that it must:
- exert a wide spectrum of antimicrobial activity at low inclusion levels.
- maintain activity throughout product manufacture, shelf life and usage.
- not compromise the quality or performance of product, pack or delivery system.
- not adversely affect patient safety or tolerance of the product.
Table 3 - Performance Requirements for Preservatives

Table 3 illustrates such requirements. Relevant microbiology textbooks provide more extensive background [2,3].
It will be evident; from the performance criteria in Table 3 that the limited list of acceptable materials is likely to be further reduced by these other considerations. Physicochemical and organoleptic properties may limit choice for some product types while possibilities for interactions with the active ingredient or excipients, the pack or delivery device must also be considered. Such properties and performance criteria could form the basis for a Target Product Profi le (TPP) with respect to the preservation system that can then be addressed in the formulation design program.
The Preservative Challenge Test (Antimicrobial Effectiveness Test)
Pharmacopoeial antimicrobial effectiveness tests (AET) or preservative efficacy tests (PET) involve challenging a product with a defi ned number of colony forming units (cfu) of a variety of test microorganisms (bacteria, yeasts and fungi), enumeration at time zero and then monitoring kill / survival rate at defi ned time intervals up to 28-days [14-16]. Test organisms that are recommended by all of the pharmacopoeias include,
- Gram positive coccus, Staphylococcus aureus.
- Gram negative rod, Pseudomonas aeruginosa.
- Fungi / mold, Aspergillus niger.
- Yeast, Candida albicans.
In addition, USP [14] and Ph. Eur. [15] recommend the use of E. coli. The list may be supplemented by additional organisms that may be associated with a particular process, facility or material, e.g. Burkholderia cepaceia an opportunistic pathogen often isolated in manufacturing environments, Bacillis subtilis a spore-forming bacteria, etc.
Acceptance criteria for USP [14] and JP [16] are broadly similar with some differences between product type and presentation. All require satisfactory reduction for each challenge organism with no subsequent increase from the initial count after 14- and 28-days. However, it is widely recognized that the criteria of the Ph. Eur. [15] are the more stringent and challenging to meet. The Ph. Eur. requires a specifi ed reduction in bacterial count within the fi rst 14-days with no subsequent increase from the initial count after 14- and 28-days. A comparison of the relative approval criteria of the USP and Ph. Eur (EP) are shown in Table 4 and 5.
Table 4 - Comparison of USP and EP Criteria for Preservative Efficacy, I) Total Viable Bacteria

Table 5 - Comparison of USP and EP Criteria for Preservative Efficacy, II) Total Viable Fungi / Yeasts
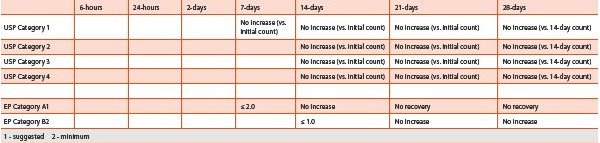
These AET tests form part of the preservative optimization studies. They also need to be performed at the end of product shelf-life to confi rm adequate preservation over the total duration of the product’s use. Some regulatory authorities also require confi rmation that the product is adequately preserved during its in-use period, when it is being routinely opened, dispensed and closed and the potential for microbial contamination is highest.
High-sensitivity test systems are being explored as possible replacements for the cumbersome, time-consuming and rather unreliable pharmacopoeial tests. Techniques include ATP bioluminescence, electrical impedance and chemiluminecence [17]. Such approaches offer the potential for automation of testing and high throughput screening of formulations during development. Inevitably, much development, validation and corroboration would be required before adopting any replacement technique. In the meantime, some issues could be addressed to ensure a more sensible approach is taken with the current monographs. These could include:
Harmonized Compendial Monographs
There is no clear evidence that the USP performance criteria have led to poorly preserved products within the US [18]. In the light of such experience and the great difficulty in getting some products to meet Ph.Eur requirements (with attendant cost and delays to product development), it would be benefi cial for the ongoing pharmacopeial harmonization initiatives to be completed as soon as possible.
Align test duration with product usage
Some oral liquid products are manufactured as lyophilized solids that are constituted with water prior to use. Shelf life in the liquid state is typically constrained by drug instability. 7 or 10 day use periods are common. Performance criteria for preservatives in such products should refl ect this. Running the test for 30 days, when the product may fail the test for other reasons e.g. loss of preservative through hydrolysis or sublimation makes little scientifi c sense. Kill / re-growth criteria should refl ect the product’s in-use shelf life.
Preservative Free Formulations
Preservative-free cosmetics, medications and food / beverages are frequently promoted as superior products. Such publication fuels demands that preservatives be omitted from medicinal products. It is true that signifi cant progress has been made in technologies for “microbiologically clean” manufacture and packaging, as well as in good manufacturing practice such that gross microbial contamination can be avoided. At the same time, many materials used as excipients, being of biological provenance cannot realistically be sterile. Furthermore, their nature or physical properties ensure that they cannot readily be sterilized before or during incorporation in product. Terminal sterilization may not be feasible for the same reasons. The presence of low levels of microbes cannot be obviated in such cases and, if the vehicle allows or encourages microbial growth (over the necessarily long shelf life that pharmaceutical products must possess) the inclusion of a preservative in the product is simply a prudent way to assure an important quality attribute and safeguard the patient.
In practical terms, removing preservative from a medicinal product (even if technically feasible) would require a comprehensive re-think of quality systems throughout manufacture to provide a product that is essentially microbe-free (essentially sterile) . Packs and delivery devices must also ensure that the quality built in at manufacture is maintained throughout the product’s shelf-life, in particular, during its in-use period. Such an approach might involve the following practices:
- Controls of microbial levels in product components (drug, excipients and especially water, container / closure) incorporating vendor certification, testing, validation of packaging and storage in appropriately clean areas.
- Product manufacture in a microbe-free environment. In some cases it may be possible to reduce or eliminate contamination by procedures during or at end of manufacture, as is possible with some food and confectionary products. However, there is a general regulatory reluctance to use preservatives to address poor manufacturing practices, to reduce viable microbial population of a non-sterile product or to control the bio-burden prior to sterilization of a multi-dose sterile product [14]. Such “post-manufacture sterilization” may deal with residual microbes but endotoxins remain and pose additional quality and safety problems.
- Product is packaged in units that maintain closure integrity during shelf life e.g. blow fill seal ampoules. Single-use units e.g. BFS are the most frequently used preservative-free container-closure systems, but are difficult to use, particularly for geriatric patients and are more costly.
Several preservation-free intranasal devices are available for commercialization [19], but there are relatively few multi-use preservative-free nasal products. It should be stressed that even using a preservative free device, the manufacturer still needs to minimize microbial contamination during manufacture / storage; there is a need to protect the nozzle during the within-use period and preventing contamination through air-intake after device actuation [19]. Strategies for preservative-free nasal devices include the use of mechanical seals, the use of embedded anti-microbial agents e.g. silver on the interior of the nozzle, filtering systems within the device to remove microbial contamination and the use of negative pressure containers to prevent ingress of micro-organisms post actuation. Some companies are developing a multi-use nasal device with self sealing nozzle that could maintain sterility after repeated microbial challenge tests [19].
Similarly, there are several novel ophthalmic container / closure systems that utilize either a 0.2 micron filter or a preservative (e.g. bezalkonium chloride) adsorbed onto a filter to maintain sterility during the in-use period, and several have been commercialized [20].
The ABAK®(Laboratoires Théa, France) and COMOD®(Ursapharm, Germany) container-closure systems have been used for several different ophthalmic products, such as common beta-blockers e.g. timolol and carteolol [21].
Technologies are generally available to assure such quality, controls and treatments being product-specific. Many pharmaceutical products already utilize such approaches. The in-use period where patients use the product still remains the Achilles heel, when the closure needs to be broached or penetrated so that a dose can be withdrawn and more importantly where there is potential for microbial contamination. There is no universally reliable way that this can be achieved for each and every product type. Getting it wrong at any stage of the supply chain, manufacture, storage or in-use will almost certainly result in a return to earlier health concerns caused by microbial contamination of preservative free multi-use ‘sterile’ medicines [22].
Conclusions
Preservatives, either singly or in synergistic combinations remain necessary to prevent microbial contamination of multi-use liquid or semi-solid medicinal products, particularly from opportunistic pathogens. Non-inclusion can result in serious patient health consequences. There are a limited number of regulatory approved preservatives that can be included in these multi-use medicinal oral or topical products and the number is constrained even further in parenteral products. Furthermore, it may be time to revisit the tests and performance requirements that products must undergo before being considered to be adequately preserved. Performance criteria and assessment techniques, based on product type, dose, environmental history in manufacture and experience during patient usage might be more appropriate than applying a single quality standard defined in pharmacopoeias that may well represent “overkill” in a microbiological and commonsense context for many products. Preservative-free approaches are still in their infancy and much more research is required before they can be considered on an equal footing with preserved approaches. However, several preservative-free intranasal and ophthalmic devices are available and do offer some promise.
Acknowledgements
Dr. Paul Newby and Dr. Don Singer, GSK for their review and comments on this manuscript.
References
- C.Moreton, Functionality and Performance of Excipients in a Quality-by-Design World, supplement to American Pharmaceutical Review, Volume 13(6): S2-S47 (2010).
- R.A. Fassihi, Preservation of Medicines against Microbial Contamination, in: S.A.Block (Ed.) Disinfection Sterilization and Preservation, 4th Edition, Lea and Febiger, 1991, pp. 871-886.
- W.B. Hugo, A.D. Russell (Eds.), Pharmaceutical Microbiology, 6th Edition, Blackwell Science, 1998, pp. 201-262 and 365-373.
- United States Pharmacopeia, USP 34-NF29, US Pharmacopeia, Rockville, Maryland, USA, 2010.
- European Pharmacopoeia EP 6.4, European Directorate for Quality of Medicines, Strasbourg, France, 2010.
- Japanese Pharmacopeia, 15th Edition, Society of Japanese Pharmacopeia, Tokyo, Japan.
- R.G.Strickley, Q.Iwata, S.Wu, T.C.Dahl, Pediatric Drugs – A review of Commercially Available Oral Formulations, J.Pharm.Sci., 97: 1731-1774 (2007).
- A.F. Fransway, The Problem of Preservation in the 1990s: III Agents with Preservation Function Independent of Formaldehyde Release, Am. J. Cont. Derm., 2: 145-174 (1991).
- B.K.Myer, A.Ni, B.Hu, L.Shi, Antimicrobial Preservative Use in Parenteral Products: Past and Present, J.Pharm.Sci., 96: 3155-3167 (2007).
- F.M. Penha, E.B. Rodrigues, M. Maia, B.A. Furlani, C. Regatieri, G.B. Melo, O. Magalhāes, R. Manzano, M.E. Farah, Retinal and Ocular Toxicity in Ocular Application of Drugs and Chemicals-Part III: Retinal Toxicity of Current and New Drugs, Ophthalmic Res., 44: 205-224 (2010).
- Draft Note for Guidance on Excipients, Antioxidants and Antimicrobial Preservatives in the Dossier for Application for Marketing Authorisation of a Medicinal Product, Committee for Proprietary Medicinal Products (CPMP), The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use, CPMP/QWP/419/03, London 20th February 2003.
- S.C. Owen, Edetic Acid Monograph, in: R.C. Rowe, P.J. Sheskey, P. J. Weller (Eds.), Handbook of Pharmaceutical Excipients, Fifth Edition, Pharmaceutical Press, 2006, pp. 260-263.
- J.Zhao, Z.Yang, M.Wang, Y.Lu, Z.Yang, Electrochemical Evaluation of the Inhibitory Effects of Weak Acids on Zagosaccharomyces baili, J.Agric. Food Chem., 52: 7246-7250 (2004).
- United States Pharmacopeia General Chapter <51> Antimicrobial Effectiveness Testing, USP 34-NF29, US Pharmacopeia, Rockville, Maryland, USA, 2010.
- European Pharmacopoeia 5.1.3 Efficacy of Antimicrobial Preservation, EP 6.4, European Directorate for Quality of Medicines, Strasbourg, France, 2010.
- Japanese Pharmacopeia, General Information: 19. Preservative Effectiveness Test, 15th Edition, Society of Japanese Pharmacopeia, Tokyo, Japan.
- P. J. Newby ‘Rapid Methods for Enumeration and Identification in Microbiology’ in Handbook of Microbiological Quality Control, Editors R.M. Baird, N.A. Hodges, S.P. Denyer, Taylor and Francis, New York, London (2000).
- S.V.W. Sutton, D. Porter, ‘Development of the Antimicrobial Effectiveness Test as USP Chaper <51>’ PDA Journal of Pharmaceutical Sciences and Technology, 56: 300-311 (2002).
- G. Brouet, Preservative-free Nasal Sprays: What Technology should be Selected and How Should it be Evaluated? Expert Opinion Biol. Ther., 3: 519-523 (2003).
- P. Furrer, J.M. Mayer, R. Gurny, Ocular Tolerance of Preservatives and Alternatives, E. J. Pharm. Biopharmac., 53: 263-280 (2002).
- C. Baudouin, A. Labbè, H. Liang, F. Brignole-Baudouin, ‘Preservatives in Eyedrops: The Good, The Bad, and The Ugly’ Progress in Retinol and Eye Research, 29: 312-334 (2010).
- L.O. Kallings, O. Ringertz, L. Silverstolpe, Microbial contamination of medical preparations, Acta Pharm. Suecica, 3: 219-228 (1996)
Author Biographies
David P. Elder has 34-years experience in the pharmaceutical industry. He is a director in the pre-clinical SCINOVO group at GSK. He has a PhD from Edinburgh University, UK. He is a member of the British Pharmacopoeia Commission and an FRSC. He has written and lectured widely on the theme of product development and the challenges of preservation.
Patrick Crowley is a pharmacist by training (FRPhSGB). He worked in the Pharmaceutical Industry for over 40 years and was a VP of product development at GSK. He currently operates as a consultant and teaches Pharmaceutical Sciences at a number of Institutions. Has authored / presented on over 40 topics related to pharmaceutical sciences.